Vaccine-Elicited Mucosal and Systemic Antibody Responses Are Associated with Reduced Simian Immunodeficiency Viremia in Infant Rhesus Macaques
- PMID: 27252535
- PMCID: PMC4984660
- DOI: 10.1128/JVI.00481-16
Vaccine-Elicited Mucosal and Systemic Antibody Responses Are Associated with Reduced Simian Immunodeficiency Viremia in Infant Rhesus Macaques
Abstract
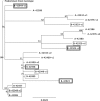
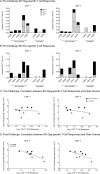
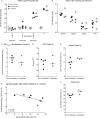
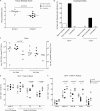
Despite significant progress in reducing peripartum mother-to-child transmission (MTCT) of human immunodeficiency virus (HIV) with antiretroviral therapy (ART), continued access to ART throughout the breastfeeding period is still a limiting factor, and breast milk exposure to HIV accounts for up to 44% of MTCT. As abstinence from breastfeeding is not recommended, alternative means are needed to prevent MTCT of HIV. We have previously shown that oral vaccination at birth with live attenuated Mycobacterium tuberculosis strains expressing simian immunodeficiency virus (SIV) genes safely induces persistent SIV-specific cellular and humoral immune responses both systemically and at the oral and intestinal mucosa. Here, we tested the ability of oral M. tuberculosis vaccine strains expressing SIV Env and Gag proteins, followed by systemic heterologous (MVA-SIV Env/Gag/Pol) boosting, to protect neonatal macaques against oral SIV challenge. While vaccination did not protect infant macaques against oral SIV acquisition, a subset of immunized animals had significantly lower peak viremia which inversely correlated with prechallenge SIV Env-specific salivary and intestinal IgA responses and higher-avidity SIV Env-specific IgG in plasma. These controller animals also maintained CD4(+) T cell populations better and showed reduced tissue pathology compared to noncontroller animals. We show that infants vaccinated at birth can develop vaccine-induced SIV-specific IgA and IgG antibodies and cellular immune responses within weeks of life. Our data further suggest that affinity maturation of vaccine-induced plasma antibodies and induction of mucosal IgA responses at potential SIV entry sites are associated with better control of viral replication, thereby likely reducing SIV morbidity.
Importance: Despite significant progress in reducing peripartum MTCT of HIV with ART, continued access to ART throughout the breastfeeding period is still a limiting factor. Breast milk exposure to HIV accounts for up to 44% of MTCT. Alternative measures, in addition to ART, are needed to achieve the goal of an AIDS-free generation. Pediatric HIV vaccines constitute a core component of such efforts. The results of our pediatric vaccine study highlight the potential importance of vaccine-elicited mucosal Env-specific IgA responses in combination with high-avidity systemic Env-specific IgG in protection against oral SIV transmission and control of viral replication in infant macaques. The induction of potent mucosal IgA antibodies by our vaccine is remarkable considering the age-dependent development of mucosal IgA responses postbirth. A deeper understanding of postnatal immune development may inform the design of improved vaccine strategies to enhance systemic and mucosal SIV/HIV antibody responses.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures




References
-
- WHO. 2014. Global update on the health sector response to HIV, 2014. World Health Organization, Geneva, Switzerland.
-
- Jensen K, Pena MG, Wilson RL, Ranganathan UD, Jacobs WR Jr, Fennelly G, Larsen M, Van Rompay KK, Kozlowski PA, Abel K. 2013. A neonatal oral-SIV prime/intramuscular MVA-SIV boost combination vaccine induces both SIV and -specific immune responses in infant macaques. Trials Vaccinol 2:53–63. doi: 10.1016/j.trivac.2013.09.005. - DOI - PMC - PubMed
-
- Jensen K, Ranganathan UD, Van Rompay KK, Canfield DR, Khan I, Ravindran R, Luciw PA, Jacobs WR Jr, Fennelly G, Larsen MH, Abel K. 2012. A recombinant attenuated Mycobacterium tuberculosis vaccine strain is safe in immunosuppressed simian immunodeficiency virus-infected infant macaques. Clin Vaccine Immunol 19:1170–1181. doi: 10.1128/CVI.00184-12. - DOI - PMC - PubMed
-
- Van Rompay KK, Abel K, Lawson JR, Singh RP, Schmidt KA, Evans T, Earl P, Harvey D, Franchini G, Tartaglia J, Montefiori D, Hattangadi S, Moss B, Marthas ML. 2005. Attenuated poxvirus-based simian immunodeficiency virus (SIV) vaccines given in infancy partially protect infant and juvenile macaques against repeated oral challenge with virulent SIV. J Acquir Immune Defic Syndr 38:124–134. doi: 10.1097/00126334-200502010-00002. - DOI - PMC - PubMed
-
- Marthas ML, Van Rompay KK, Abbott Z, Earl P, Buonocore-Buzzelli L, Moss B, Rose NF, Rose JK, Kozlowski PA, Abel K. 2011. Partial efficacy of a VSV-SIV/MVA-SIV vaccine regimen against oral SIV challenge in infant macaques. Vaccine 29:3124–3137. doi: 10.1016/j.vaccine.2011.02.051. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- F30 AA024374/AA/NIAAA NIH HHS/United States
- P51 OD011107/OD/NIH HHS/United States
- P51 RR000169/RR/NCRR NIH HHS/United States
- U01 AI068636/AI/NIAID NIH HHS/United States
- P30 CA016086/CA/NCI NIH HHS/United States
- HHSN261200800001C/RC/CCR NIH HHS/United States
- HHSN261200800001E/CA/NCI NIH HHS/United States
- T32 AI007001/AI/NIAID NIH HHS/United States
- T32 AI007419/AI/NIAID NIH HHS/United States
- R24 RR016038/RR/NCRR NIH HHS/United States
- R37 AI026170/AI/NIAID NIH HHS/United States
- P30 AI050410/AI/NIAID NIH HHS/United States
- R01 DE019064/DE/NIDCR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

