From the Cover: Disease-Induced Disparities in Formation of the Nanoparticle-Biocorona and the Toxicological Consequences
- PMID: 27255384
- PMCID: PMC4960912
- DOI: 10.1093/toxsci/kfw097
From the Cover: Disease-Induced Disparities in Formation of the Nanoparticle-Biocorona and the Toxicological Consequences
Abstract
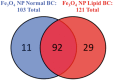
Nanoparticle (NP) association with macromolecules in a physiological environment forms a biocorona (BC), which alters NP distribution, activity, and toxicity. While BC formation is dependent on NP physicochemical properties, little information exists on the influence of the physiological environment. Obese individuals and those with cardiovascular disease exist with altered serum chemistry, which is expected to influence BC formation and NP toxicity. We hypothesize that a BC formed on NPs following incubation in hyperlipidemic serum will result in altered NP-BC protein content, cellular association, and toxicity compared to normal serum conditions. We utilized Fe3O4 NPs, which are being developed as MRI contrast and tumor targeting agents to test our hypothesis. We used rat aortic endothelial cells (RAECs) within a dynamic flow in vitro exposure system to more accurately depict the in vivo environment. A BC was formed on 20nm PVP-suspended Fe3O4 NPs following incubation in water, 10% normal or hyperlipidemic rat serum. Addition of BCs resulted in increased hydrodynamic size and decreased surface charge. More cholesterol associated with Fe3O4 NPs after incubation in hyperlipidemic as compared with normal serum. Using quantitative proteomics, we identified unique differences in BC protein components between the 2 serum types. Under flow conditions, formation of a BC from both serum types reduced RAECs association of Fe3O4 NPs. Addition of BCs was found to exacerbate RAECs inflammatory gene responses to Fe3O4 NPs (Fe3O4-hyperlipidemic > Fe3O4-normal > Fe3O4) including increased expression of IL-6, TNF-α, Cxcl-2, VCAM-1, and ICAM-1. Overall, these findings demonstrate that disease-induced variations in physiological environments have a significant impact NP-BC formation, cellular association, and cell response.
Keywords: darkfield microscopy; endothelial cell; hyperlipidemia; iron oxide nanoparticles; nanotoxicology; protein corona.; proteomics.
© The Author 2016. Published by Oxford University Press on behalf of the Society of Toxicology. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Figures




Similar articles
-
Altered formation of the iron oxide nanoparticle-biocorona due to individual variability and exercise.Environ Toxicol Pharmacol. 2018 Sep;62:215-226. doi: 10.1016/j.etap.2018.07.014. Epub 2018 Jul 29. Environ Toxicol Pharmacol. 2018. PMID: 30096581 Free PMC article.
-
Experimental challenges regarding the in vitro investigation of the nanoparticle-biocorona in disease states.Toxicol In Vitro. 2018 Sep;51:40-49. doi: 10.1016/j.tiv.2018.05.003. Epub 2018 May 5. Toxicol In Vitro. 2018. PMID: 29738787 Free PMC article.
-
Modulation of Pulmonary Toxicity in Metabolic Syndrome Due to Variations in Iron Oxide Nanoparticle-Biocorona Composition.Nanomaterials (Basel). 2022 Jun 11;12(12):2022. doi: 10.3390/nano12122022. Nanomaterials (Basel). 2022. PMID: 35745361 Free PMC article.
-
Surface engineering of inorganic nanoparticles for imaging and therapy.Adv Drug Deliv Rev. 2013 May;65(5):622-48. doi: 10.1016/j.addr.2012.08.015. Epub 2012 Sep 6. Adv Drug Deliv Rev. 2013. PMID: 22975010 Review.
-
Biocorona-induced modifications in engineered nanomaterial-cellular interactions impacting biomedical applications.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2020 May;12(3):e1608. doi: 10.1002/wnan.1608. Epub 2019 Dec 1. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2020. PMID: 31788989 Free PMC article. Review.
Cited by
-
Altered formation of the iron oxide nanoparticle-biocorona due to individual variability and exercise.Environ Toxicol Pharmacol. 2018 Sep;62:215-226. doi: 10.1016/j.etap.2018.07.014. Epub 2018 Jul 29. Environ Toxicol Pharmacol. 2018. PMID: 30096581 Free PMC article.
-
Formation of a protein corona influences the biological identity of nanomaterials.Rep Pract Oncol Radiother. 2018 Jul-Aug;23(4):300-308. doi: 10.1016/j.rpor.2018.05.005. Epub 2018 May 28. Rep Pract Oncol Radiother. 2018. PMID: 30100819 Free PMC article.
-
The biocorona: a challenge for the biomedical application of nanoparticles.Nanotechnol Rev. 2017 Aug;6(4):345-353. doi: 10.1515/ntrev-2016-0098. Epub 2017 Jan 20. Nanotechnol Rev. 2017. PMID: 29607287 Free PMC article.
-
Experimental challenges regarding the in vitro investigation of the nanoparticle-biocorona in disease states.Toxicol In Vitro. 2018 Sep;51:40-49. doi: 10.1016/j.tiv.2018.05.003. Epub 2018 May 5. Toxicol In Vitro. 2018. PMID: 29738787 Free PMC article.
-
Protective Effect of Silver Nanoparticles Against Cytosine Arabinoside Genotoxicity: An In Vivo Micronucleus Assay.Int J Environ Res Public Health. 2024 Dec 18;21(12):1689. doi: 10.3390/ijerph21121689. Int J Environ Res Public Health. 2024. PMID: 39767527 Free PMC article.
References
-
- Aldossari A. A., Shannahan J. H., Podila R., Brown J. M. (2015b). Scavenger receptor B1 facilitates macrophage uptake of silver nanoparticles and cellular activation. J Nanopart Res 17, 313.
-
- Amiri H., Bordonali L., Lascialfari A., Wan S., Monopoli M. P., Lynch I., Laurent S., Mahmoudi M. (2013). Protein corona affects the relaxivity and MRI contrast efficiency of magnetic nanoparticles. Nanoscale 5, 8656–8665. - PubMed
-
- Babes L., Denizot B., Tanguy G., Le Jeune J. J., Jallet P. (1999). Synthesis of iron oxide nanoparticles used as MRI contrast agents: a parametric study. J Colloid Interface Sci. 212, 474–482. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

