Transcriptomic and molecular genetic analysis of the cell wall salvage response of Aspergillus niger to the absence of galactofuranose synthesis
- PMID: 27264789
- PMCID: PMC5129474
- DOI: 10.1111/cmi.12624
Transcriptomic and molecular genetic analysis of the cell wall salvage response of Aspergillus niger to the absence of galactofuranose synthesis
Abstract
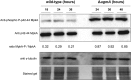
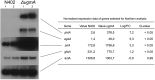
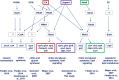
The biosynthesis of cell surface-located galactofuranose (Galf)-containing glycostructures such as galactomannan, N-glycans and O-glycans in filamentous fungi is important to secure the integrity of the cell wall. UgmA encodes an UDP-galactopyranose mutase, which is essential for the formation of Galf. Consequently, the ΔugmA mutant lacks Galf-containing molecules. Our previous work in Aspergillus niger work suggested that loss of function of ugmA results in activation of the cell wall integrity (CWI) pathway which is characterized by increased expression of the agsA gene, encoding an α-glucan synthase. In this study, the transcriptional response of the ΔugmA mutant was further linked to the CWI pathway by showing the induced and constitutive phosphorylation of the CWI-MAP kinase in the ΔugmA mutant. To identify genes involved in cell wall remodelling in response to the absence of galactofuranose biosynthesis, a genome-wide expression analysis was performed using RNAseq. Over 400 genes were higher expressed in the ΔugmA mutant compared to the wild-type. These include genes that encode enzymes involved in chitin (gfaB, gnsA, chsA) and α-glucan synthesis (agsA), and in β-glucan remodelling (bgxA, gelF and dfgC), and also include several glycosylphosphatidylinositol (GPI)-anchored cell wall protein-encoding genes. In silico analysis of the 1-kb promoter regions of the up-regulated genes in the ΔugmA mutant indicated overrepresentation of genes with RlmA, MsnA, PacC and SteA-binding sites. The importance of these transcription factors for survival of the ΔugmA mutant was analysed by constructing the respective double mutants. The ΔugmA/ΔrlmA and ΔugmA/ΔmsnA double mutants showed strong synthetic growth defects, indicating the importance of these transcription factors to maintain cell wall integrity in the absence of Galf biosynthesis.
© 2016 The Authors Cellular Microbiology Published by John Wiley & Sons Ltd.
Figures



Similar articles
-
UDP-Galactopyranose Mutase Mediates Cell Wall Integrity, Polarity Growth, and Virulence in Fusarium graminearum.Appl Environ Microbiol. 2023 Feb 28;89(2):e0123522. doi: 10.1128/aem.01235-22. Epub 2023 Jan 19. Appl Environ Microbiol. 2023. PMID: 36656025 Free PMC article.
-
Identification of the UDP-glucose-4-epimerase required for galactofuranose biosynthesis and galactose metabolism in A. niger.Fungal Biol Biotechnol. 2014 Oct 14;1:6. doi: 10.1186/s40694-014-0006-7. eCollection 2014. Fungal Biol Biotechnol. 2014. PMID: 28955448 Free PMC article.
-
Identification and functional analysis of two Golgi-localized UDP-galactofuranose transporters with overlapping functions in Aspergillus niger.BMC Microbiol. 2015 Nov 2;15:253. doi: 10.1186/s12866-015-0541-2. BMC Microbiol. 2015. PMID: 26526354 Free PMC article.
-
Biosynthesis of galactomannans found in filamentous fungi belonging to Pezizomycotina.Biosci Biotechnol Biochem. 2018 Feb;82(2):183-191. doi: 10.1080/09168451.2017.1422383. Epub 2018 Jan 15. Biosci Biotechnol Biochem. 2018. PMID: 29334321 Review.
-
GPI Anchored Proteins in Aspergillus fumigatus and Cell Wall Morphogenesis.Curr Top Microbiol Immunol. 2020;425:167-186. doi: 10.1007/82_2020_207. Curr Top Microbiol Immunol. 2020. PMID: 32418035 Review.
Cited by
-
Updating genome annotation for the microbial cell factory Aspergillus niger using gene co-expression networks.Nucleic Acids Res. 2019 Jan 25;47(2):559-569. doi: 10.1093/nar/gky1183. Nucleic Acids Res. 2019. PMID: 30496528 Free PMC article.
-
Identification of chitin synthase activator in Aspergillus niger and its application in citric acid fermentation.Appl Microbiol Biotechnol. 2022 Nov;106(21):6993-7011. doi: 10.1007/s00253-022-12174-9. Epub 2022 Sep 23. Appl Microbiol Biotechnol. 2022. PMID: 36149454
-
Conidium Specific Polysaccharides in Aspergillus fumigatus.J Fungi (Basel). 2023 Jan 24;9(2):155. doi: 10.3390/jof9020155. J Fungi (Basel). 2023. PMID: 36836270 Free PMC article.
-
Identification of galactofuranose antigens such as galactomannoproteins and fungal-type galactomannan from the yellow koji fungus (Aspergillus oryzae).Front Microbiol. 2023 Feb 6;14:1110996. doi: 10.3389/fmicb.2023.1110996. eCollection 2023. Front Microbiol. 2023. PMID: 36814571 Free PMC article.
-
The α-(1,3)-glucan synthase gene agsE impacts the secretome of Aspergillus niger.Antonie Van Leeuwenhoek. 2023 Sep;116(9):867-882. doi: 10.1007/s10482-023-01853-w. Epub 2023 Jun 14. Antonie Van Leeuwenhoek. 2023. PMID: 37316742 Free PMC article.
References
-
- Afroz, S. , El‐Ganiny, A.M. , Sanders, D.A. , and Kaminskyj, S.G. (2011) Roles of the Aspergillus nidulans UDP‐galactofuranose transporter, UgtA in hyphal morphogenesis, cell wall architecture, conidiation, and drug sensitivity. Fungal Genet Biol 48: 896–903. - PubMed
-
- Alam, M.K. , El‐Ganiny, A.M. , Afroz, S. , Sanders, D.A.R. , Liu, J. , and Kaminskyj, S.G.W. (2012) Aspergillus nidulans galactofuranose biosynthesis affects antifungal drug sensitivity. Fungal Genet Biol 49: 1033–1043. - PubMed
-
- Alexander, B.D. , and Pfaller, M.A. (2006) Contemporary Tools for the Diagnosis and Management of Invasive Mycoses. Clin Infect Dis 43: S15–S27.
-
- Arentshorst, M. , Jing, N. , and Ram, A.F. (2015) Efficient generation of Aspergillus niger knock out strains by combining NHEJ mutants and a split marker approach In Genetic Transformation Systems in Fungi, Volume 1. van den Berg M.A., Maruthachalam K, (eds). Switzerland: Springer International Publishing, pp. 263–272.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

