Effectiveness of Sofosbuvir, Ledipasvir/Sofosbuvir, or Paritaprevir/Ritonavir/Ombitasvir and Dasabuvir Regimens for Treatment of Patients With Hepatitis C in the Veterans Affairs National Health Care System
- PMID: 27267053
- PMCID: PMC5341745
- DOI: 10.1053/j.gastro.2016.05.049
Effectiveness of Sofosbuvir, Ledipasvir/Sofosbuvir, or Paritaprevir/Ritonavir/Ombitasvir and Dasabuvir Regimens for Treatment of Patients With Hepatitis C in the Veterans Affairs National Health Care System
Erratum in
-
Correction.Gastroenterology. 2016 Dec;151(6):1253. doi: 10.1053/j.gastro.2016.11.006. Epub 2016 Nov 14. Gastroenterology. 2016. PMID: 27855272 No abstract available.
Abstract
Background & aims: We investigated the real-world effectiveness of sofosbuvir, ledipasvir/sofosbuvir, and paritaprevir/ritonavir/ombitasvir and dasabuvir (PrOD) in treatment of different subgroups of patients infected with hepatitis C virus (HCV) genotypes 1, 2, 3, or 4.
Methods: We performed a retrospective analysis of data from 17,487 patients with HCV infection (13,974 with HCV genotype 1; 2131 with genotype 2; 1237 with genotype 3; and 135 with genotype 4) who began treatment with sofosbuvir (n = 2986), ledipasvir/sofosbuvir (n = 11,327), or PrOD (n = 3174), with or without ribavirin, from January 1, 2014 through June 20, 2015 in the Veterans Affairs health care system. Data through April 15, 2016 were analyzed to assess completion of treatments and sustained virologic response 12 weeks after treatment (SVR12). Mean age of patients was 61 ± 7 years, 97% were male, 52% were non-Hispanic white, 29% were non-Hispanic black, 32% had a diagnosis of cirrhosis (9.9% with decompensated cirrhosis), 36% had a Fibrosis-4 index score >3.25 (indicator of cirrhosis), and 29% had received prior antiviral treatment.
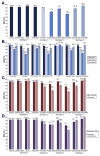
Results: An SVR12 was achieved by 92.8% (95% confidence interval [CI], 92.3%-93.2%) of subjects with HCV genotype 1 infection (no significant difference between ledipasvir/sofosbuvir and PrOD regimens), 86.2% (95% CI, 84.6%-87.7%) of those with genotype 2 infection (treated with sofosbuvir and ribavirin), 74.8% (95% CI, 72.2%-77.3%) of those with genotype 3 infection (77.9% in patients given ledipasvir/sofosbuvir plus ribavirin, 87.0% in patients given sofosbuvir and pegylated-interferon plus ribavirin, and 70.6% of patients given sofosbuvir plus ribavirin), and 89.6% (95% CI 82.8%-93.9%) of those with genotype 4 infection. Among patients with cirrhosis, 90.6% of patients with HCV genotype 1, 77.3% with HCV genotype 2, 65.7% with HCV genotype 3, and 83.9% with HCV genotype 4 achieved an SVR12. Among previously treated patients, 92.6% with genotype 1; 80.2% with genotype 2; 69.2% with genotype 3; and 93.5% with genotype 4 achieved SVR12. Among treatment-naive patients, 92.8% with genotype 1; 88.0% with genotype 2; 77.5% with genotype 3; and 88.3% with genotype 4 achieved SVR12. Eight-week regimens of ledipasvir/sofosbuvir produced an SVR12 in 94.3% of eligible patients with HCV genotype 1 infection; this regimen was underused.
Conclusions: High proportions of patients with HCV infections genotypes 1-4 (ranging from 75% to 93%) in the Veterans Affairs national health care system achieved SVR12, approaching the results reported in clinical trials, especially in patients with genotype 1 infection. An 8-week regimen of ledipasvir/sofosbuvir is effective for eligible patients with HCV genotype 1 infection and could reduce costs. There is substantial room for improvement in SVRs among persons with cirrhosis and genotype 2 or 3 infections.
Keywords: DAA; Harvoni; VA; Viekira.
Copyright © 2016 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors disclose no conflicts.
Figures

Comment in
-
"New" Cancer Genes and Inherited Colorectal Cancer Risk: Caveat Emptor.Gastroenterology. 2017 Jan;152(1):12-13. doi: 10.1053/j.gastro.2016.11.027. Epub 2016 Nov 23. Gastroenterology. 2017. PMID: 27889571 No abstract available.
References
-
- Andreone P, Colombo MG, Enejosa JV, et al. ABT-450, ritonavir, ombitasvir, and dasabuvir achieves 97% and 100% sustained virologic response with or without ribavirin in treatment-experienced patients with HCV genotype 1b infection. Gastroenterology. 2014;147:359–365 e1. - PubMed
-
- Ferenci P, Bernstein D, Lalezari J, et al. ABT-450/r-ombitasvir and dasabuvir with or without ribavirin for HCV. N Engl J Med. 2014;370:1983–1992. - PubMed
-
- Kowdley KV, Gordon SC, Reddy KR, et al. Ledipasvir and sofosbuvir for 8 or 12 weeks for chronic HCV without cirrhosis. N Engl J Med. 2014;370:1879–1888. - PubMed
-
- Poordad F, Hezode C, Trinh R, et al. ABT-450/r-ombitasvir and dasabuvir with ribavirin for hepatitis C with cirrhosis. N Engl J Med. 2014;370:1973–1982. - PubMed
-
- Zeuzem S, Jacobson IM, Baykal T, et al. Retreatment of HCV with ABT-450/r-ombitasvir and dasabuvir with ribavirin. N Engl J Med. 2014;370:1604–1614. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

