Steps toward improving ethical evaluation in health technology assessment: a proposed framework
- PMID: 27267369
- PMCID: PMC4895959
- DOI: 10.1186/s12910-016-0118-0
Steps toward improving ethical evaluation in health technology assessment: a proposed framework
Abstract
Background: While evaluation of ethical aspects in health technology assessment (HTA) has gained much attention during the past years, the integration of ethics in HTA practice still presents many challenges. In response to the increasing demand for expansion of health technology assessment (HTA) methodology to include ethical issues more systematically, this article reports on a multi-stage study that aimed at construction of a framework for improving the integration of ethics in HTA.
Methods: The framework was developed through the following phases: 1) a systematic review and content analysis of guidance documents for ethics in HTA; 2) identification of factors influencing the integration of ethical considerations in HTA; 3) preparation of an action-oriented framework based on the key elements of the existing guidance documents and identified barriers to and facilitators of their implementation; and 4) expert consultation and revision of the framework.
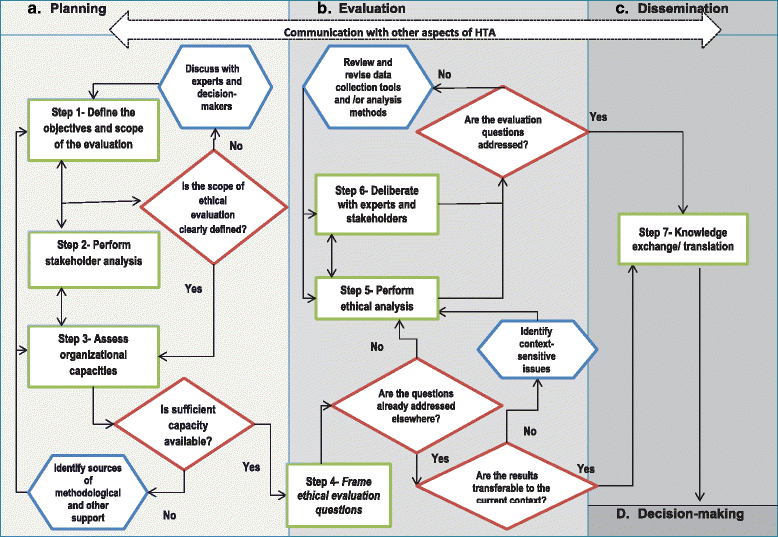
Results: The proposed framework consists of three main components: an algorithmic flowchart, which exhibits the different steps of an ethical inquiry throughout the HTA process, including: defining the objectives and scope of the evaluation, stakeholder analysis, assessing organizational capacity, framing ethical evaluation questions, ethical analysis, deliberation, and knowledge translation; a stepwise guide, which focuses on the task objectives and potential questions that are required to be addressed at each step; and a list of some commonly recommended or used tools to help facilitate the evaluation process.
Conclusions: The proposed framework can be used to support and promote good practice in integration of ethics into HTA. However, further validation of the framework through case studies and expert consultation is required to establish its utility for HTA practice.
Keywords: Ethics; Framework; Health technology assessment; Model; Tools.
Figures
Similar articles
-
Barriers and facilitators influencing ethical evaluation in health technology assessment.Int J Technol Assess Health Care. 2015 Jan;31(3):113-23. doi: 10.1017/S026646231500032X. Int J Technol Assess Health Care. 2015. PMID: 26179275
-
Integrating ethics in health technology assessment: many ways to Rome.Int J Technol Assess Health Care. 2015 Jan;31(3):131-7. doi: 10.1017/S0266462315000276. Epub 2015 Jun 11. Int J Technol Assess Health Care. 2015. PMID: 26063128
-
Evaluation of Ethical Analyses in Seven Reports from the European Network for Health Technology Assessment.Int J Technol Assess Health Care. 2019;35(4):273-279. doi: 10.1017/S0266462319000485. Epub 2019 Jul 16. Int J Technol Assess Health Care. 2019. PMID: 31307559 Free PMC article.
-
Ethics in Canadian health technology assessment: a descriptive review.Int J Technol Assess Health Care. 2009 Oct;25(4):463-9. doi: 10.1017/S0266462309990390. Int J Technol Assess Health Care. 2009. PMID: 19845976 Review.
-
ETHICS IN HEALTH TECHNOLOGY ASSESSMENT: A SYSTEMATIC REVIEW.Int J Technol Assess Health Care. 2018 Jan;34(5):447-457. doi: 10.1017/S0266462318000508. Epub 2018 Oct 9. Int J Technol Assess Health Care. 2018. PMID: 30296950
Cited by
-
Proactive and Ongoing Analysis and Management of Ethical Concerns in the Development, Evaluation, and Implementation of Smart Homes for Older Adults With Frailty.JMIR Aging. 2023 Mar 9;6:e41322. doi: 10.2196/41322. JMIR Aging. 2023. PMID: 36892912 Free PMC article.
-
Targeting ethical considerations tied to image-based mobile health diagnostic support specific to clinicians in low-resource settings: the Brocher proposition.Glob Health Action. 2019;12(1):1666695. doi: 10.1080/16549716.2019.1666695. Glob Health Action. 2019. PMID: 31532350 Free PMC article.
-
Introducing responsible innovation in health: a policy-oriented framework.Health Res Policy Syst. 2018 Sep 10;16(1):90. doi: 10.1186/s12961-018-0362-5. Health Res Policy Syst. 2018. PMID: 30200985 Free PMC article.
-
Seven pillars for ethics in digital diagnostic assistance among clinicians: Take-homes from a multi-stakeholder and multi-country workshop.J Glob Health. 2020 Jun;10(1):010326. doi: 10.7189/jogh.10.010326. J Glob Health. 2020. PMID: 32509282 Free PMC article. No abstract available.
-
Good Practices for Health Technology Assessment Guideline Development: A Report of the Health Technology Assessment International, HTAsiaLink, and ISPOR Special Task Force.Int J Technol Assess Health Care. 2025 Jan 6;40(1):e74. doi: 10.1017/S0266462324004719. Int J Technol Assess Health Care. 2025. PMID: 39760423 Free PMC article.
References
-
- European Network for Health Technology Assessment (EUnetHTA) HTA Core Model® online, Version 2.1 edn. Helsinki: National Institute for Health and Welfare; 2014.
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous