Hyperthermia treatment of tumors by mesenchymal stem cell-delivered superparamagnetic iron oxide nanoparticles
- PMID: 27274229
- PMCID: PMC4869665
- DOI: 10.2147/IJN.S94255
Hyperthermia treatment of tumors by mesenchymal stem cell-delivered superparamagnetic iron oxide nanoparticles
Abstract
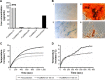
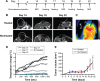
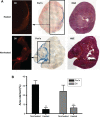
Magnetic hyperthermia - a potential cancer treatment in which superparamagnetic iron oxide nanoparticles (SPIONs) are made to resonantly respond to an alternating magnetic field (AMF) and thereby produce heat - is of significant current interest. We have previously shown that mesenchymal stem cells (MSCs) can be labeled with SPIONs with no effect on cell proliferation or survival and that within an hour of systemic administration, they migrate to and integrate into tumors in vivo. Here, we report on some longer term (up to 3 weeks) post-integration characteristics of magnetically labeled human MSCs in an immunocompromized mouse model. We initially assessed how the size and coating of SPIONs dictated the loading capacity and cellular heating of MSCs. Ferucarbotran(®) was the best of those tested, having the best like-for-like heating capability and being the only one to retain that capability after cell internalization. A mouse model was created by subcutaneous flank injection of a combination of 0.5 million Ferucarbotran-loaded MSCs and 1.0 million OVCAR-3 ovarian tumor cells. After 2 weeks, the tumors reached ~100 µL in volume and then entered a rapid growth phase over the third week to reach ~300 µL. In the control mice that received no AMF treatment, magnetic resonance imaging (MRI) data showed that the labeled MSCs were both incorporated into and retained within the tumors over the entire 3-week period. In the AMF-treated mice, heat increases of ~4°C were observed during the first application, after which MRI indicated a loss of negative contrast, suggesting that the MSCs had died and been cleared from the tumor. This post-AMF removal of cells was confirmed by histological examination and also by a reduced level of subsequent magnetic heating effect. Despite this evidence for an AMF-elicited response in the SPION-loaded MSCs, and in contrast to previous reports on tumor remission in immunocompetent mouse models, in this case, no significant differences were measured regarding the overall tumor size or growth characteristics. We discuss the implications of these results on the clinical delivery of hyperthermia therapy to tumors and on the possibility that a preferred therapeutic route may involve AMF as an adjuvant to an autologous immune response.
Keywords: MRI; SPIONs; hyperthermia; mesenchymal stem cells; tumor therapy.
Figures
References
-
- Hildebrandt B, Wust P, Ahlers O, et al. The cellular and molecular basis of hyperthermia. Crit Rev Oncol Hematol. 2002;43(1):33–56. - PubMed
-
- Cavaliere R, Ciocatto EC, Giovanella BC, et al. Selective heat sensitivity of cancer cells. Biochemical and clinical studies. Cancer. 1967;20(9):1351–1381. - PubMed
-
- Jordan A, Scholz R, Maier-Hauff K, et al. Presentation of a new magnetic field therapy system for the treatment of human solid tumors with magnetic fluid hyperthermia. J Magn Magn Mater. 2001;225(1–2):118–126.
-
- Thomas LA, Dekker L, Kallumadil M, et al. Carboxylic acid-stabilised iron oxide nanoparticles for use in magnetic hyperthermia. J Mater Chem. 2009;19(36):6529–6535.
-
- Hiergeist R, Andra W, Buske N, et al. Application of magnetite ferrofluids for hyperthermia. J Magn Magn Mater. 1999;201:420–422.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

