Actomyosin Cortical Mechanical Properties in Nonadherent Cells Determined by Atomic Force Microscopy
- PMID: 27276270
- PMCID: PMC4906360
- DOI: 10.1016/j.bpj.2016.04.034
Actomyosin Cortical Mechanical Properties in Nonadherent Cells Determined by Atomic Force Microscopy
Abstract
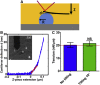
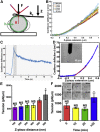
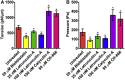
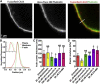
The organization of filamentous actin and myosin II molecular motor contractility is known to modify the mechanical properties of the cell cortical actomyosin cytoskeleton. Here we describe a novel method, to our knowledge, for using force spectroscopy approach curves with tipless cantilevers to determine the actomyosin cortical tension, elastic modulus, and intracellular pressure of nonadherent cells. We validated the method by measuring the surface tension of water in oil microdrops deposited on a glass surface. We extracted an average tension of T ∼ 20.25 nN/μm, which agrees with macroscopic experimental methods. We then measured cortical mechanical properties in nonadherent human foreskin fibroblasts and THP-1 human monocytes before and after pharmacological perturbations of actomyosin activity. Our results show that myosin II activity and actin polymerization increase cortex tension and intracellular pressure, whereas branched actin networks decreased them. Interestingly, myosin II activity stiffens the cortex and branched actin networks soften it, but actin polymerization has no effect on cortex stiffness. Our method is capable of detecting changes in cell mechanical properties in response to perturbations of the cytoskeleton, allowing characterization with physically relevant parameters. Altogether, this simple method should be of broad application for deciphering the molecular regulation of cell cortical mechanical properties.
Published by Elsevier Inc.
Figures


References
-
- Salbreux G., Charras G., Paluch E. Actin cortex mechanics and cellular morphogenesis. Trends Cell Biol. 2012;22:536–545. - PubMed
-
- Clark A.G., Wartlick O., Paluch E.K. Stresses at the cell surface during animal cell morphogenesis. Curr. Biol. 2014;24:R484–R494. - PubMed
-
- Charras G., Paluch E. Blebs lead the way: how to migrate without lamellipodia. Nat. Rev. Mol. Cell Biol. 2008;9:730–736. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

