Membrane Tension Acts Through PLD2 and mTORC2 to Limit Actin Network Assembly During Neutrophil Migration
- PMID: 27280401
- PMCID: PMC4900667
- DOI: 10.1371/journal.pbio.1002474
Membrane Tension Acts Through PLD2 and mTORC2 to Limit Actin Network Assembly During Neutrophil Migration
Abstract
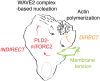
For efficient polarity and migration, cells need to regulate the magnitude and spatial distribution of actin assembly. This process is coordinated by reciprocal interactions between the actin cytoskeleton and mechanical forces. Actin polymerization-based protrusion increases tension in the plasma membrane, which in turn acts as a long-range inhibitor of actin assembly. These interactions form a negative feedback circuit that limits the magnitude of membrane tension in neutrophils and prevents expansion of the existing front and the formation of secondary fronts. It has been suggested that the plasma membrane directly inhibits actin assembly by serving as a physical barrier that opposes protrusion. Here we show that efficient control of actin polymerization-based protrusion requires an additional mechanosensory feedback cascade that indirectly links membrane tension with actin assembly. Specifically, elevated membrane tension acts through phospholipase D2 (PLD2) and the mammalian target of rapamycin complex 2 (mTORC2) to limit actin nucleation. In the absence of this pathway, neutrophils exhibit larger leading edges, higher membrane tension, and profoundly defective chemotaxis. Mathematical modeling suggests roles for both the direct (mechanical) and indirect (biochemical via PLD2 and mTORC2) feedback loops in organizing cell polarity and motility-the indirect loop is better suited to enable competition between fronts, whereas the direct loop helps spatially organize actin nucleation for efficient leading edge formation and cell movement. This circuit is essential for polarity, motility, and the control of membrane tension.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Mammalian target of rapamycin and Rictor control neutrophil chemotaxis by regulating Rac/Cdc42 activity and the actin cytoskeleton.Mol Biol Cell. 2013 Nov;24(21):3369-80. doi: 10.1091/mbc.E13-07-0405. Epub 2013 Sep 4. Mol Biol Cell. 2013. PMID: 24006489 Free PMC article.
-
Mechanosensitive mTORC2 independently coordinates leading and trailing edge polarity programs during neutrophil migration.Mol Biol Cell. 2023 May 1;34(5):ar35. doi: 10.1091/mbc.E22-05-0191. Epub 2023 Mar 1. Mol Biol Cell. 2023. PMID: 36857159 Free PMC article.
-
Endothelial Cell mTOR Complex-2 Regulates Sprouting Angiogenesis.PLoS One. 2015 Aug 21;10(8):e0135245. doi: 10.1371/journal.pone.0135245. eCollection 2015. PLoS One. 2015. PMID: 26295809 Free PMC article.
-
Ras-related GTPases and the cytoskeleton.Mol Biol Cell. 1992 May;3(5):475-9. doi: 10.1091/mbc.3.5.475. Mol Biol Cell. 1992. PMID: 1611153 Free PMC article. Review.
-
Actin dynamics at the leading edge: from simple machinery to complex networks.Dev Cell. 2009 Sep;17(3):310-22. doi: 10.1016/j.devcel.2009.08.012. Dev Cell. 2009. PMID: 19758556 Review.
Cited by
-
Cortical tension promotes Kibra degradation via Par-1.Mol Biol Cell. 2024 Jan 1;35(1):ar2. doi: 10.1091/mbc.E23-06-0246. Epub 2023 Oct 30. Mol Biol Cell. 2024. PMID: 37903240 Free PMC article.
-
Whole-genome screens reveal regulators of differentiation state and context-dependent migration in human neutrophils.Nat Commun. 2023 Sep 18;14(1):5770. doi: 10.1038/s41467-023-41452-x. Nat Commun. 2023. PMID: 37723145 Free PMC article.
-
Optogenetic control of receptors reveals distinct roles for actin- and Cdc42-dependent negative signals in chemotactic signal processing.Nat Commun. 2021 Nov 16;12(1):6148. doi: 10.1038/s41467-021-26371-z. Nat Commun. 2021. PMID: 34785668 Free PMC article.
-
Mechanoreciprocity in cell migration.Nat Cell Biol. 2018 Jan;20(1):8-20. doi: 10.1038/s41556-017-0012-0. Epub 2017 Dec 21. Nat Cell Biol. 2018. PMID: 29269951 Free PMC article. Review.
-
Microtubules control cellular shape and coherence in amoeboid migrating cells.J Cell Biol. 2020 Jun 1;219(6):e201907154. doi: 10.1083/jcb.201907154. J Cell Biol. 2020. PMID: 32379884 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

