LASS5 Interacts with SDHB and Synergistically Represses p53 and p21 Activity
- PMID: 27280497
- PMCID: PMC5280074
- DOI: 10.2174/1566524016666160607090012
LASS5 Interacts with SDHB and Synergistically Represses p53 and p21 Activity
Abstract
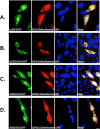
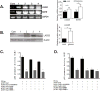
Longevity Assurance 5 (LASS5), a member of the LASS/Ceramide Synthases family, synthesizes C16-ceramide and is implicated in tumor biology. However, its precise role is not yet well understood. A yeast two-hybrid screen was performed using a human cDNA library to identify potential LASS5- interaction partners. One identified clone encodes succinate dehydrogenase subunit B (SDHB). Mammalian two-hybrid assays showed that LASS5 interacts with SDHB, and the result was also confirmed by GST pull-down and coimmunoprecipitation assays. The C-terminal fragment of SDHB was required for the interaction. LASS5 and SDHB were co-localized in COS-7 cells. LASS5 and SDHB expressions were found to be up-regulated in neuroglioma tissue. Transfection assays showed that LASS5 or SDHB expression repressed p53 or p21 reporter activity, respectively. Simultaneous LASS5 and SDHB expression resulted in stronger repression of p53 and p21 reporter activity, suggesting that LASS5 and SDHB interaction may synergistically affect transcriptional regulation of p53 and p21. Our data provide new molecular insights into potential roles of LASS5 and SDHB in tumor biology.
Conflict of interest statement
The authors confirm that this article content has no conflicts of interest.
Figures

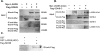
Similar articles
-
Synergistic efficacy of LBH and alphaB-crystallin through inhibiting transcriptional activities of p53 and p21.BMB Rep. 2010 Jun;43(6):432-7. doi: 10.5483/bmbrep.2010.43.6.432. BMB Rep. 2010. PMID: 20587334
-
Modulation of the cyclin-dependent kinase inhibitor p21(WAF1/Cip1) gene by Zac1 through the antagonistic regulators p53 and histone deacetylase 1 in HeLa Cells.Mol Cancer Res. 2008 Jul;6(7):1204-14. doi: 10.1158/1541-7786.MCR-08-0123. Mol Cancer Res. 2008. PMID: 18644983
-
Indirect p53-dependent transcriptional repression of Survivin, CDC25C, and PLK1 genes requires the cyclin-dependent kinase inhibitor p21/CDKN1A and CDE/CHR promoter sites binding the DREAM complex.Oncotarget. 2015 Dec 8;6(39):41402-17. doi: 10.18632/oncotarget.6356. Oncotarget. 2015. PMID: 26595675 Free PMC article.
-
Reduced p21(WAF1/CIP1) via alteration of p53-DDX3 pathway is associated with poor relapse-free survival in early-stage human papillomavirus-associated lung cancer.Clin Cancer Res. 2011 Apr 1;17(7):1895-905. doi: 10.1158/1078-0432.CCR-10-2316. Epub 2011 Feb 16. Clin Cancer Res. 2011. PMID: 21325288
-
PBK/TOPK interacts with the DBD domain of tumor suppressor p53 and modulates expression of transcriptional targets including p21.Oncogene. 2010 Oct 7;29(40):5464-74. doi: 10.1038/onc.2010.275. Epub 2010 Jul 12. Oncogene. 2010. PMID: 20622899
Cited by
-
Natural Products and Small Molecules Targeting Cellular Ceramide Metabolism to Enhance Apoptosis in Cancer Cells.Cancers (Basel). 2023 Sep 20;15(18):4645. doi: 10.3390/cancers15184645. Cancers (Basel). 2023. PMID: 37760612 Free PMC article. Review.
-
Astrocytic ceramide as possible indicator of neuroinflammation.J Neuroinflammation. 2019 Feb 25;16(1):48. doi: 10.1186/s12974-019-1436-1. J Neuroinflammation. 2019. PMID: 30803453 Free PMC article.
-
C16-ceramide is a natural regulatory ligand of p53 in cellular stress response.Nat Commun. 2018 Oct 8;9(1):4149. doi: 10.1038/s41467-018-06650-y. Nat Commun. 2018. PMID: 30297838 Free PMC article.
-
High expression of ceramide synthase 5 predicts a poor prognosis in gastric cancer.Transl Cancer Res. 2022 Sep;11(9):3209-3221. doi: 10.21037/tcr-22-1348. Transl Cancer Res. 2022. PMID: 36237237 Free PMC article.
References
-
- Lahiri S, Futerman AH. LASS5 is a bona fide dihydroceramide synthase that selectively utilizes palmitoyl CoA as acyl donor. J Biol Chem. 2005;280:33735–8. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
