Biguanides sensitize leukemia cells to ABT-737-induced apoptosis by inhibiting mitochondrial electron transport
- PMID: 27283492
- PMCID: PMC5239486
- DOI: 10.18632/oncotarget.9843
Biguanides sensitize leukemia cells to ABT-737-induced apoptosis by inhibiting mitochondrial electron transport
Abstract
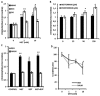
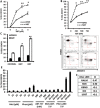
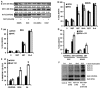
Metformin displays antileukemic effects partly due to activation of AMPK and subsequent inhibition of mTOR signaling. Nevertheless, Metformin also inhibits mitochondrial electron transport at complex I in an AMPK-independent manner, Here we report that Metformin and rotenone inhibit mitochondrial electron transport and increase triglyceride levels in leukemia cell lines, suggesting impairment of fatty acid oxidation (FAO). We also report that, like other FAO inhibitors, both agents and the related biguanide, Phenformin, increase sensitivity to apoptosis induction by the bcl-2 inhibitor ABT-737 supporting the notion that electron transport antagonizes activation of the intrinsic apoptosis pathway in leukemia cells. Both biguanides and rotenone induce superoxide generation in leukemia cells, indicating that oxidative damage may sensitize toABT-737 induced apoptosis. In addition, we demonstrate that Metformin sensitizes leukemia cells to the oligomerization of Bak, suggesting that the observed synergy with ABT-737 is mediated, at least in part, by enhanced outer mitochondrial membrane permeabilization. Notably, Phenformin was at least 10-fold more potent than Metformin in abrogating electron transport and increasing sensitivity to ABT-737, suggesting that this agent may be better suited for targeting hematological malignancies. Taken together, our results suggest that inhibition of mitochondrial metabolism by Metformin or Phenformin is associated with increased leukemia cell susceptibility to induction of intrinsic apoptosis, and provide a rationale for clinical studies exploring the efficacy of combining biguanides with the orally bioavailable derivative of ABT-737, Venetoclax.
Keywords: ABT 737; leukemia; metformin; mitochondria; phenformin.
Conflict of interest statement
The authors declare that they have not conflict of interest.
Figures



Similar articles
-
Norcantharidin enhances ABT-737-induced apoptosis in hepatocellular carcinoma cells by transcriptional repression of Mcl-1.Cell Signal. 2012 Sep;24(9):1803-9. doi: 10.1016/j.cellsig.2012.05.009. Epub 2012 May 17. Cell Signal. 2012. PMID: 22609455
-
Clitocine induces apoptosis and enhances the lethality of ABT-737 in human colon cancer cells by disrupting the interaction of Mcl-1 and Bak.Cancer Lett. 2014 Dec 28;355(2):253-63. doi: 10.1016/j.canlet.2014.09.024. Epub 2014 Oct 7. Cancer Lett. 2014. PMID: 25304383
-
Methyl succinate antagonises biguanide-induced AMPK-activation and death of pancreatic beta-cells through restoration of mitochondrial electron transfer.Br J Pharmacol. 2007 Apr;150(8):1031-43. doi: 10.1038/sj.bjp.0707189. Epub 2007 Mar 5. Br J Pharmacol. 2007. PMID: 17339833 Free PMC article.
-
Therapeutic Repurposing of Biguanides in Cancer.Trends Cancer. 2021 Aug;7(8):714-730. doi: 10.1016/j.trecan.2021.03.001. Epub 2021 Apr 14. Trends Cancer. 2021. PMID: 33865798 Free PMC article. Review.
-
Targeting the Electron Transport System for Enhanced Longevity.Biomolecules. 2025 Apr 23;15(5):614. doi: 10.3390/biom15050614. Biomolecules. 2025. PMID: 40427507 Free PMC article. Review.
Cited by
-
Exploiting Mitochondrial Vulnerabilities to Trigger Apoptosis Selectively in Cancer Cells.Cancers (Basel). 2019 Jun 29;11(7):916. doi: 10.3390/cancers11070916. Cancers (Basel). 2019. PMID: 31261935 Free PMC article. Review.
-
Novel Mango Ginger Bioactive (2,4,6-Trihydroxy-3,5-diprenyldihydrochalcone) Inhibits Mitochondrial Metabolism in Combination with Avocatin B.ACS Omega. 2022 Jan 7;7(2):1682-1693. doi: 10.1021/acsomega.1c04053. eCollection 2022 Jan 18. ACS Omega. 2022. PMID: 35071863 Free PMC article.
-
Nitric Oxide-Mediated Enhancement and Reversal of Resistance of Anticancer Therapies.Antioxidants (Basel). 2019 Sep 17;8(9):407. doi: 10.3390/antiox8090407. Antioxidants (Basel). 2019. PMID: 31533363 Free PMC article. Review.
-
Disarib, a Specific BCL2 Inhibitor, Induces Apoptosis in Triple-Negative Breast Cancer Cells and Impedes Tumour Progression in Xenografts by Altering Mitochondria-Associated Processes.Int J Mol Sci. 2024 Jun 12;25(12):6485. doi: 10.3390/ijms25126485. Int J Mol Sci. 2024. PMID: 38928195 Free PMC article.
-
Metformin use and cervical cancer risk in female patients with type 2 diabetes.Oncotarget. 2016 Sep 13;7(37):59548-59555. doi: 10.18632/oncotarget.10934. Oncotarget. 2016. PMID: 27486978 Free PMC article.
References
-
- Ofran Y, Rowe JM. Treatment for relapsed acute myeloid leukemia: what is new? Curr Opin Hematol. 2012;19:89–94. - PubMed
-
- Narayanan S, Shami PJ. Treatment of acute lymphoblastic leukemia in adults. Crit Rev Oncol Hematol. 2012;81:94–102. - PubMed
-
- Konopleva M, Andreeff M. Targeting the leukemia microenvironment. Curr Drug Targets. 2007;8:685–701. - PubMed
-
- Tabe Y, Konopleva M, Igari J, Andreeff M. Spontaneous migration of acute promyelocytic leukemia cells beneath cultured bone marrow adipocytes with matched expression of the major histocompatibility complex. Rinsho Byori. 2004;52:642–648. - PubMed
-
- Ishikawa F, Yoshida S, Saito Y, Hijikata A, Kitamura H, Tanaka S, Nakamura R, Tanaka T, Tomiyama H, Saito N, Fukata M, Miyamoto T, Lyons B, Ohshima K, Uchida N, Taniguchi S, et al. Chemotherapy-resistant human AML stem cells home to and engraft within the bone-marrow endosteal region. Nat Biotechnol. 2007;25:1315–1321. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

