Alk1 controls arterial endothelial cell migration in lumenized vessels
- PMID: 27287800
- PMCID: PMC4958337
- DOI: 10.1242/dev.135392
Alk1 controls arterial endothelial cell migration in lumenized vessels
Abstract
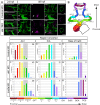
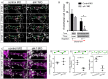
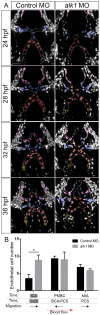
Heterozygous loss of the arterial-specific TGFβ type I receptor, activin receptor-like kinase 1 (ALK1; ACVRL1), causes hereditary hemorrhagic telangiectasia (HHT). HHT is characterized by development of fragile, direct connections between arteries and veins, or arteriovenous malformations (AVMs). However, how decreased ALK1 signaling leads to AVMs is unknown. To understand the cellular mis-steps that cause AVMs, we assessed endothelial cell behavior in alk1-deficient zebrafish embryos, which develop cranial AVMs. Our data demonstrate that alk1 loss has no effect on arterial endothelial cell proliferation but alters arterial endothelial cell migration within lumenized vessels. In wild-type embryos, alk1-positive cranial arterial endothelial cells generally migrate towards the heart, against the direction of blood flow, with some cells incorporating into endocardium. In alk1-deficient embryos, migration against flow is dampened and migration in the direction of flow is enhanced. Altered migration results in decreased endothelial cell number in arterial segments proximal to the heart and increased endothelial cell number in arterial segments distal to the heart. We speculate that the consequent increase in distal arterial caliber and hemodynamic load precipitates the flow-dependent development of downstream AVMs.
Keywords: Alk1/Acvrl1; Angiogenesis; Arteriovenous malformation; Endocardium; Hereditary hemorrhagic telangiectasia; Zebrafish.
© 2016. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures



References
-
- Chen H., Brady Ridgway J., Sai T., Lai J., Warming S., Chen H., Roose-Girma M., Zhang G., Shou W. and Yan M. (2013). Context-dependent signaling defines roles of BMP9 and BMP10 in embryonic and postnatal development. Proc. Natl. Acad. Sci. USA 110, 11887-11892. 10.1073/pnas.1306074110 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

