Luteolin inhibits GABAA receptors in HEK cells and brain slices
- PMID: 27292079
- PMCID: PMC4904371
- DOI: 10.1038/srep27695
Luteolin inhibits GABAA receptors in HEK cells and brain slices
Abstract
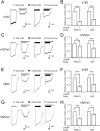
Modulation of the A type γ-aminobutyric acid receptors (GABAAR) is one of the major drug targets for neurological and psychological diseases. The natural flavonoid compound luteolin (2-(3,4-Dihydroxyphenyl)- 5,7-dihydroxy-4-chromenone) has been reported to have antidepressant, antinociceptive, and anxiolytic-like effects, which possibly involve the mechanisms of modulating GABA signaling. However, as yet detailed studies of the pharmacological effects of luteolin are still lacking, we investigated the effects of luteolin on recombinant and endogenous GABAAR-mediated current responses by electrophysiological approaches. Our results showed that luteolin inhibited GABA-mediated currents and slowed the activation kinetics of recombinant α1β2, α1β2γ2, α5β2, and α5β2γ2 receptors with different degrees of potency and efficacy. The modulatory effect of luteolin was likely dependent on the subunit composition of the receptor complex: the αβ receptors were more sensitive than the αβγ receptors. In hippocampal pyramidal neurons, luteolin significantly reduced the amplitude and slowed the rise time of miniature inhibitory postsynaptic currents (mIPSCs). However, GABAAR-mediated tonic currents were not significantly influenced by luteolin. These data suggested that luteolin has negative modulatory effects on both recombinant and endogenous GABAARs and inhibits phasic rather than tonic inhibition in hippocampus.
Figures





Similar articles
-
Physiological and pharmacological properties of inhibitory postsynaptic currents mediated by α5β1γ2, α5β2γ2 and α5β3γ2 GABAA receptors.Neuropharmacology. 2017 Oct;125:243-253. doi: 10.1016/j.neuropharm.2017.07.027. Epub 2017 Jul 27. Neuropharmacology. 2017. PMID: 28757051
-
Etomidate blocks LTP and impairs learning but does not enhance tonic inhibition in mice carrying the N265M point mutation in the beta3 subunit of the GABA(A) receptor.Neuropharmacology. 2015 Jun;93:171-178. doi: 10.1016/j.neuropharm.2015.01.011. Epub 2015 Feb 11. Neuropharmacology. 2015. PMID: 25680234 Free PMC article.
-
Inhibition of a tonic inhibitory conductance in mouse hippocampal neurones by negative allosteric modulators of α5 subunit-containing γ-aminobutyric acid type A receptors: implications for treating cognitive deficits.Br J Anaesth. 2021 Mar;126(3):674-683. doi: 10.1016/j.bja.2020.11.032. Epub 2020 Dec 30. Br J Anaesth. 2021. PMID: 33388140
-
Molecular and Regulatory Mechanisms of Desensitization and Resensitization of GABAA Receptors with a Special Reference to Propofol/Barbiturate.Int J Mol Sci. 2020 Jan 15;21(2):563. doi: 10.3390/ijms21020563. Int J Mol Sci. 2020. PMID: 31952324 Free PMC article. Review.
-
An Emerging Circuit Pharmacology of GABAA Receptors.Trends Pharmacol Sci. 2018 Aug;39(8):710-732. doi: 10.1016/j.tips.2018.04.003. Epub 2018 Jun 11. Trends Pharmacol Sci. 2018. PMID: 29903580 Free PMC article. Review.
Cited by
-
Antidepressant and anxiolytic activity of Lavandula officinalis aerial parts hydroalcoholic extract in scopolamine-treated rats.Pharm Biol. 2017 Dec;55(1):958-965. doi: 10.1080/13880209.2017.1285320. Pharm Biol. 2017. PMID: 28166686 Free PMC article.
-
Flumazenil-Insensitive Benzodiazepine Effects in Recombinant αβ and Neuronal GABAA Receptors.Brain Sci. 2020 Mar 5;10(3):150. doi: 10.3390/brainsci10030150. Brain Sci. 2020. PMID: 32150806 Free PMC article.
-
Phenolics as GABAA Receptor Ligands: An Updated Review.Molecules. 2022 Mar 8;27(6):1770. doi: 10.3390/molecules27061770. Molecules. 2022. PMID: 35335130 Free PMC article. Review.
-
The Neuroprotective Effect of Rooibos Herbal Tea Against Alzheimer's Disease: A Review.Mol Nutr Food Res. 2025 Jan;69(1):e202400670. doi: 10.1002/mnfr.202400670. Epub 2024 Dec 19. Mol Nutr Food Res. 2025. PMID: 39703045 Free PMC article. Review.
-
Luteolin reversed anxiety and depressive-like behavior via modulation of the NF-κB/NLRP3 inflammasome axis in the hippocampus of rats subjected to sleep deprivation.Iran J Basic Med Sci. 2024;27(8):1050-1058. doi: 10.22038/IJBMS.2024.75068.16277. Iran J Basic Med Sci. 2024. PMID: 38911248 Free PMC article.
References
-
- Miean K. H. & Mohamed S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. Journal of agricultural and food chemistry 49, 3106–3112 (2001). - PubMed
-
- Lopez-Lazaro M. Distribution and biological activities of the flavonoid luteolin. Mini reviews in medicinal chemistry 9, 31–59 (2009). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

