Inexpensive, serotype-independent protocol for native and bioengineered recombinant adeno-associated virus purification
- PMID: 27294171
- PMCID: PMC4902285
- DOI: 10.14440/jbm.2016.102
Inexpensive, serotype-independent protocol for native and bioengineered recombinant adeno-associated virus purification
Abstract
Recombinant adeno-associated virus (AAV) is a valuable and often used gene therapy vector. With increased demand for highly purified virus comes the need for a standardized purification procedure that is applicable across many serotypes and includes bioengineered viruses. Currently cesium chloride banding or affinity chromatography are the predominate forms of purification. These approaches expose the final purified virus to toxic contaminants or are highly capsid dependent and may require significant optimization to isolate purified AAV. These methods may also limit crude viral lysate processing volume resulting in a significant loss of viral titer. To circumvent these issues, we have developed an AAV purification protocol independent of toxic compounds, supernatant volume and capsid moiety. This purification method standardizes virus purification across native serotype and bioengineered mosaic capsids.
Keywords: PEG8000; adeno-associated virus; centrifugation; inexpensive; purification.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

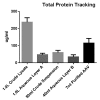

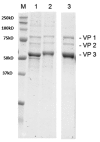
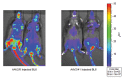

References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
