Insomnia, Health-Related Quality of Life and Health Outcomes in Children: A Seven Year Longitudinal Cohort
- PMID: 27295263
- PMCID: PMC4904740
- DOI: 10.1038/srep27921
Insomnia, Health-Related Quality of Life and Health Outcomes in Children: A Seven Year Longitudinal Cohort
Abstract
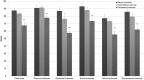
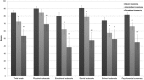
Insomnia is common in children, and is associated with decreased school performance and increased psychopathology. Although adult insomnia is linked to worsened health-related quality of life (HRQOL), there is insufficient data evaluating insomnia and HRQOL in children. We examined the HRQOL and health associations of insomnia in a longitudinal cohort of 194 children (96 girls, age at study start 8.7 ± 1.6 years, age at data analysis 15.0 ± 1.8 years) over 7 years. International Classification of Sleep Disorders, second edition (ICSD2) derived insomnia was seen intermittently in 27% of children, and was persistent in 4%. Children reporting ICSD2-derived insomnia had lower HRQOL. Additionally, the presence of insomnia was associated with an increased risk of reporting a new medical condition (intermittent insomnia odds ratio 5.9 [95% CI 1.3-26.7, p = 0.04], persistent insomnia odds ratio 8 [95% CI 2.3-27.7, p = 0.001]). Persistent ICSD2-derived insomnia was associated with an increased risk of reporting a new medication (odds ratio 4.9 (95% CI 1.0-23.6), p = 0.049), and reporting a new psychiatric medication (odds ratio 13.7, 95% CI: 2.6-73.5, p = 0.002). These associations were present even after adjusting for socioeconomic factors and the presence of obstructive sleep apnea. Insomnia in children is associated with worsened HRQOL and health outcomes.
Conflict of interest statement
Dr. Parthasarathy reports grants from NIH/NHLBI, grants from Patient Centered Outcomes Research Institute, grants from US Department of Defense, grants from NIH (National Cancer Institute) NCI, grants from US Department of Army, grants from Johrei Institute, personal fees from American Academy of Sleep Medicine, personal fees from American College of Chest Physicians, personal fees from UpToDate Inc., personal fees from Philips-Respironics, Inc. and Vapotherm Inc., grants from Younes Sleep Technologies, Ltd., grants from Niveus Medical Inc., grants from Philips-Respironics, Inc., outside the submitted work. Dr. Parthasarathy reports non-financial support from National Center for Sleep Disorders Research of the NIH (NHLBI) and patents UA 14-018 U.S.S.N. 61/884,654; PTAS 502570970 (Home breathing device) pending. Dr. Wayne Morgan has received grant funding from NIH and the Cystic Fibrosis Foundation and is a consultant to the Cystic Fibrosis Foundation and Genentech. These are all unrelated to the topic of this paper. Dr. Stuart F. Quan reports grants from the NIH/NIA, American Diabetes Association, and the National Science Foundation, and consults for Global Corporate Challenge and the American Board of Internal Medicine.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

