The Juvenile Phase of Maize Sees Upregulation of Stress-Response Genes and Is Extended by Exogenous Jasmonic Acid
- PMID: 27307257
- PMCID: PMC4972259
- DOI: 10.1104/pp.15.01707
The Juvenile Phase of Maize Sees Upregulation of Stress-Response Genes and Is Extended by Exogenous Jasmonic Acid
Erratum in
-
CORRECTION: Vol. 171: 2648-2658, 2016.Plant Physiol. 2017 Feb;173(2):1519. doi: 10.1104/pp.16.01922. Plant Physiol. 2017. PMID: 28154113 Free PMC article. No abstract available.
Abstract
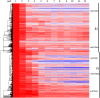
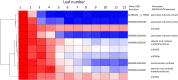
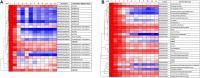
As maize (Zea mays) plants undergo vegetative phase change from juvenile to adult, they both exhibit heteroblasty, an abrupt change in patterns of leaf morphogenesis, and gain the ability to produce flowers. Both processes are under the control of microRNA156 (miR156), whose levels decline at the end of the juvenile phase. Gain of the ability to flower is conferred by the expression of miR156 targets that encode SQUAMOSA PROMOTER-BINDING transcription factors, which, when derepressed in the adult phase, induce the expression of MADS box transcription factors that promote maturation and flowering. How gene expression, including targets of those microRNAs, differs between the two phases remains an open question. Here, we compare transcript levels in primordia that will develop into juvenile or adult leaves to identify genes that define these two developmental states and may influence vegetative phase change. In comparisons among successive leaves at the same developmental stage, plastochron 6, three-fourths of approximately 1,100 differentially expressed genes were more highly expressed in primordia of juvenile leaves. This juvenile set was enriched in photosynthetic genes, particularly those associated with cyclic electron flow at photosystem I, and in genes involved in oxidative stress and retrograde redox signaling. Pathogen- and herbivory-responsive pathways including salicylic acid and jasmonic acid also were up-regulated in juvenile primordia; indeed, exogenous application of jasmonic acid delayed both the appearance of adult traits and the decline in the expression of miR156-encoding loci in maize seedlings. We hypothesize that the stresses associated with germination promote juvenile patterns of differentiation in maize.
© 2016 American Society of Plant Biologists. All Rights Reserved.
Figures




Similar articles
-
Induction of annexin by heavy metals and jasmonic acid in Zea mays.Funct Integr Genomics. 2013 Jun;13(2):241-51. doi: 10.1007/s10142-013-0316-5. Epub 2013 Mar 9. Funct Integr Genomics. 2013. PMID: 23474989
-
Microarray analysis of vegetative phase change in maize.Plant J. 2008 Dec;56(6):1045-57. doi: 10.1111/j.1365-313X.2008.03661.x. Epub 2008 Sep 19. Plant J. 2008. PMID: 18764925
-
Root herbivory: molecular analysis of the maize transcriptome upon infestation by Southern corn rootworm, Diabrotica undecimpunctata howardi.Physiol Plant. 2012 Apr;144(4):303-19. doi: 10.1111/j.1399-3054.2011.01557.x. Epub 2012 Jan 23. Physiol Plant. 2012. PMID: 22172013
-
The integration of leaf-derived signals sets the timing of vegetative phase change in maize, a process coordinated by epigenetic remodeling.Plant Sci. 2021 Nov;312:111035. doi: 10.1016/j.plantsci.2021.111035. Epub 2021 Aug 27. Plant Sci. 2021. PMID: 34620439 Review.
-
Juvenile Leaves or Adult Leaves: Determinants for Vegetative Phase Change in Flowering Plants.Int J Mol Sci. 2020 Dec 21;21(24):9753. doi: 10.3390/ijms21249753. Int J Mol Sci. 2020. PMID: 33371265 Free PMC article. Review.
Cited by
-
QTL and candidate genes for heterophylly in soybean based on two populations of recombinant inbred lines.Front Plant Sci. 2022 Aug 16;13:961619. doi: 10.3389/fpls.2022.961619. eCollection 2022. Front Plant Sci. 2022. PMID: 36051289 Free PMC article.
-
The Maize MID-COMPLEMENTING ACTIVITY Homolog CELL NUMBER REGULATOR13/NARROW ODD DWARF Coordinates Organ Growth and Tissue Patterning.Plant Cell. 2017 Mar;29(3):474-490. doi: 10.1105/tpc.16.00878. Epub 2017 Mar 2. Plant Cell. 2017. PMID: 28254777 Free PMC article.
-
Cytokinin depends on GA biosynthesis and signaling to regulate different aspects of vegetative phase change in Arabidopsis.Nat Commun. 2025 Jul 8;16(1):6292. doi: 10.1038/s41467-025-61507-5. Nat Commun. 2025. PMID: 40628766 Free PMC article.
-
RNA-directed DNA methylation prevents rapid and heritable reversal of transposon silencing under heat stress in Zea mays.PLoS Genet. 2021 Jun 14;17(6):e1009326. doi: 10.1371/journal.pgen.1009326. eCollection 2021 Jun. PLoS Genet. 2021. PMID: 34125827 Free PMC article.
-
Coordinated regulation of vegetative phase change by brassinosteroids and the age pathway in Arabidopsis.Nat Commun. 2023 May 5;14(1):2608. doi: 10.1038/s41467-023-38207-z. Nat Commun. 2023. PMID: 37147280 Free PMC article.
References
-
- Achard P, Renou JP, Berthomé R, Harberd NP, Genschik P (2008) Plant DELLAs restrain growth and promote survival of adversity by reducing the levels of reactive oxygen species. Curr Biol 18: 656–660 - PubMed
-
- Acosta IF, Laparra H, Romero SP, Schmelz E, Hamberg M, Mottinger JP, Moreno MA, Dellaporta SL (2009) tasselseed1 is a lipoxygenase affecting jasmonic acid signaling in sex determination of maize. Science 323: 262–265 - PubMed
-
- Baena-González E, Rolland F, Thevelein JM, Sheen J (2007) A central integrator of transcription networks in plant stress and energy signalling. Nature 23: 938–942 - PubMed
-
- Bailey T, Elkan C (1994) Fitting a mixture model by expectation maximization to discover motifs in biopolymers. Proc Int Conf Intell Syst Mol Biol 2: 28–36 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

