Traffic jams and the complex role of α-Synuclein aggregation in Parkinson disease
- PMID: 27314512
- PMCID: PMC5464113
- DOI: 10.1080/21541248.2016.1199191
Traffic jams and the complex role of α-Synuclein aggregation in Parkinson disease
Abstract
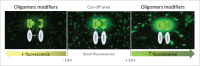
A common pathological event among various neurodegenerative disorders (NDs) is the misfolding and aggregation of different proteins in the brain. This is thought to potentiate aberrant protein-protein interactions that culminate in the disruption of several biological processes and, ultimately, in neuronal cell loss. Although protein aggregates are a common hallmark in several disorders, the molecular pathways leading to their generation remain unclear. The misfolding and aggregation of α-Synuclein (aSyn) is the pathological hallmark of Parkinson disease (PD), the second most common age related ND. It has been postulated that oligomeric species of aSyn, rather than more mature aggregated forms of the protein, are the causative agents of cytotoxicity. In recent years, we have been investigating the molecular mechanisms underlying the initial steps of aSyn accumulation in living cells. Using an unbiased genome-wide lentiviral RNAi screen we identified trafficking and kinase genes as modulators of aSyn oligomerization, aggregation, and toxicity. Among those, Rab8b, Rab11a, Rab13 and Slp5 were found to promote the clearance of aSyn inclusions and reduce aSyn toxicity. Moreover, we found that endocytic recycling and secretion of aSyn was enhanced upon expression of Rab11a or Rab13 in cells accumulating aSyn inclusions. Altogether, our findings suggest specific trafficking steps may prove beneficial as targets for therapeutic intervention in synucleinopathies, and should be further investigated in other models.
Keywords: Parkinson disease; Rab GTPases; Slp5; aggregation; neuronal trafficking; oligomerization; α-Synuclein.
Figures
Similar articles
-
shRNA-Based Screen Identifies Endocytic Recycling Pathway Components That Act as Genetic Modifiers of Alpha-Synuclein Aggregation, Secretion and Toxicity.PLoS Genet. 2016 Apr 28;12(4):e1005995. doi: 10.1371/journal.pgen.1005995. eCollection 2016 Apr. PLoS Genet. 2016. PMID: 27123591 Free PMC article.
-
Systematic comparison of the effects of alpha-synuclein mutations on its oligomerization and aggregation.PLoS Genet. 2014 Nov 13;10(11):e1004741. doi: 10.1371/journal.pgen.1004741. eCollection 2014 Nov. PLoS Genet. 2014. PMID: 25393002 Free PMC article.
-
The effects of the novel A53E alpha-synuclein mutation on its oligomerization and aggregation.Acta Neuropathol Commun. 2016 Dec 9;4(1):128. doi: 10.1186/s40478-016-0402-8. Acta Neuropathol Commun. 2016. PMID: 27938414 Free PMC article.
-
α-Synuclein Pathology in Synucleinopathies: Mechanisms, Biomarkers, and Therapeutic Challenges.Int J Mol Sci. 2025 Jun 4;26(11):5405. doi: 10.3390/ijms26115405. Int J Mol Sci. 2025. PMID: 40508212 Free PMC article. Review.
-
Oxidative and nitrative alpha-synuclein modifications and proteostatic stress: implications for disease mechanisms and interventions in synucleinopathies.J Neurochem. 2013 May;125(4):491-511. doi: 10.1111/jnc.12226. Epub 2013 Mar 19. J Neurochem. 2013. PMID: 23452040 Review.
Cited by
-
Unveiling sex-based differences in Parkinson's disease: a comprehensive meta-analysis of transcriptomic studies.Biol Sex Differ. 2022 Nov 22;13(1):68. doi: 10.1186/s13293-022-00477-5. Biol Sex Differ. 2022. PMID: 36414996 Free PMC article.
-
α-Synuclein Trafficking in Parkinson's Disease: Insights From Fly and Mouse Models.ASN Neuro. 2018 Jan-Dec;10:1759091418812587. doi: 10.1177/1759091418812587. ASN Neuro. 2018. PMID: 30482039 Free PMC article. Review.
-
Synuclein impairs trafficking and signaling of BDNF in a mouse model of Parkinson's disease.Sci Rep. 2017 Jun 20;7(1):3868. doi: 10.1038/s41598-017-04232-4. Sci Rep. 2017. PMID: 28634349 Free PMC article.
-
Overexpression of alpha synuclein disrupts APP and Endolysosomal axonal trafficking in a mouse model of synucleinopathy.Neurobiol Dis. 2023 Mar;178:106010. doi: 10.1016/j.nbd.2023.106010. Epub 2023 Jan 23. Neurobiol Dis. 2023. PMID: 36702318 Free PMC article.
-
Single Molecule Fingerprinting Reveals Different Amplification Properties of α-Synuclein Oligomers and Preformed Fibrils in Seeding Assay.ACS Chem Neurosci. 2022 Apr 6;13(7):883-896. doi: 10.1021/acschemneuro.1c00553. Epub 2022 Mar 14. ACS Chem Neurosci. 2022. PMID: 35286811 Free PMC article.
References
-
- Spillantini MG, Crowther RA, Jakes R, Cairns NJ, Lantos PL, Goedert M. Filamentous alpha-synuclein inclusions link multiple system atrophy with Parkinson's disease and dementia with Lewy bodies. Neurosci Lett 1998; 251(3):205-8; PMID:9726379; https://doi.org/10.1016/S0304-3940(98)00504-7 - DOI - PubMed
-
- Spillantini MG, Crowther RA, Jakes R, Hasegawa M, Goedert M. alpha-Synuclein in filamentous inclusions of Lewy bodies from Parkinson's disease and dementia with lewy bodies. Proc Natl Acad Sci U S A 1998; 95(11):6469-73; PMID:9600990; https://doi.org/10.1073/pnas.95.11.6469 - DOI - PMC - PubMed
-
- Outeiro TF, Putcha P, Tetzlaff JE, Spoelgen R, Koker M, Carvalho F, Hyman BT, McLean PJ. Formation of toxic oligomeric alpha-synuclein species in living cells. PLoS One 2008; 3(4):e1867; PMID:18382657; https://doi.org/10.1371/journal.pone.0001867 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
