Neutrophil-Derived IL-1β Impairs the Efficacy of NF-κB Inhibitors against Lung Cancer
- PMID: 27320908
- PMCID: PMC4927403
- DOI: 10.1016/j.celrep.2016.05.085
Neutrophil-Derived IL-1β Impairs the Efficacy of NF-κB Inhibitors against Lung Cancer
Abstract
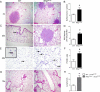
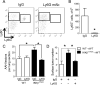
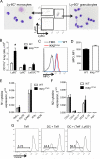
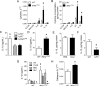
Although epithelial NF-κB signaling is important for lung carcinogenesis, NF-κB inhibitors are ineffective for cancer treatment. To explain this paradox, we studied mice with genetic deletion of IKKβ in myeloid cells and found enhanced tumorigenesis in Kras(G12D) and urethane models of lung cancer. Myeloid-specific inhibition of NF-κB augmented pro-IL-1β processing by cathepsin G in neutrophils, leading to increased IL-1β and enhanced epithelial cell proliferation. Combined treatment with bortezomib, a proteasome inhibitor that blocks NF-κB activation, and IL-1 receptor antagonist reduced tumor formation and growth in vivo. In lung cancer patients, plasma IL-1β levels correlated with poor prognosis, and IL-1β increased following bortezomib treatment. Together, our studies elucidate an important role for neutrophils and IL-1β in lung carcinogenesis and resistance to NF-κB inhibitors.
Copyright © 2016 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
NF-kappaB is a negative regulator of IL-1beta secretion as revealed by genetic and pharmacological inhibition of IKKbeta.Cell. 2007 Sep 7;130(5):918-31. doi: 10.1016/j.cell.2007.07.009. Cell. 2007. PMID: 17803913 Free PMC article.
-
Cell-selective inhibition of NF-κB signaling improves therapeutic index in a melanoma chemotherapy model.Cancer Discov. 2011 Nov;1(6):496-507. doi: 10.1158/2159-8290.CD-11-0143. Cancer Discov. 2011. PMID: 22389871 Free PMC article.
-
Opposing effects of bortezomib-induced nuclear factor-κB inhibition on chemical lung carcinogenesis.Carcinogenesis. 2012 Apr;33(4):859-67. doi: 10.1093/carcin/bgs024. Epub 2012 Jan 27. Carcinogenesis. 2012. PMID: 22287559 Free PMC article.
-
NF-kappaB in lung cancer, a carcinogenesis mediator and a prevention and therapy target.Front Biosci (Landmark Ed). 2011 Jan 1;16(3):1172-85. doi: 10.2741/3782. Front Biosci (Landmark Ed). 2011. PMID: 21196225 Free PMC article. Review.
-
The Fire Within: NF-κB Involvement in Non-Small Cell Lung Cancer.Cancer Res. 2020 Oct 1;80(19):4025-4036. doi: 10.1158/0008-5472.CAN-19-3578. Epub 2020 Jul 2. Cancer Res. 2020. PMID: 32616502 Review.
Cited by
-
Neutrophil dynamics in the tumor microenvironment.J Clin Invest. 2021 Mar 15;131(6):e143759. doi: 10.1172/JCI143759. J Clin Invest. 2021. PMID: 33720040 Free PMC article. Review.
-
KRAS-Mutant Lung Cancer: Targeting Molecular and Immunologic Pathways, Therapeutic Advantages and Restrictions.Cells. 2023 Feb 26;12(5):749. doi: 10.3390/cells12050749. Cells. 2023. PMID: 36899885 Free PMC article. Review.
-
Making cold malignant pleural effusions hot: driving novel immunotherapies.Oncoimmunology. 2019 Jan 22;8(4):e1554969. doi: 10.1080/2162402X.2018.1554969. eCollection 2019. Oncoimmunology. 2019. PMID: 30906651 Free PMC article. Review.
-
The Roles of Inflammasomes in Host Defense against Mycobacterium tuberculosis.Pathogens. 2021 Jan 25;10(2):120. doi: 10.3390/pathogens10020120. Pathogens. 2021. PMID: 33503864 Free PMC article. Review.
-
Polymorphisms within Autophagy-Related Genes Influence the Risk of Developing Colorectal Cancer: A Meta-Analysis of Four Large Cohorts.Cancers (Basel). 2021 Mar 12;13(6):1258. doi: 10.3390/cancers13061258. Cancers (Basel). 2021. PMID: 33809172 Free PMC article.
References
-
- Besse B, Planchard D, Veillard AS, Taillade L, Khayat D, Ducourtieux M, Pignon JP, Lumbroso J, Lafontaine C, Mathiot C, Soria JC. Phase 2 study of frontline bortezomib in patients with advanced non-small cell lung cancer. Lung Cancer. 2012;76:78–83. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

