Matrix metalloproteinases in the brain and blood-brain barrier: Versatile breakers and makers
- PMID: 27323783
- PMCID: PMC5012524
- DOI: 10.1177/0271678X16655551
Matrix metalloproteinases in the brain and blood-brain barrier: Versatile breakers and makers
Abstract
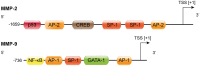
Matrix metalloproteinases are versatile endopeptidases with many different functions in the body in health and disease. In the brain, matrix metalloproteinases are critical for tissue formation, neuronal network remodeling, and blood-brain barrier integrity. Many reviews have been published on matrix metalloproteinases before, most of which focus on the two best studied matrix metalloproteinases, the gelatinases MMP-2 and MMP-9, and their role in one or two diseases. In this review, we provide a broad overview of the role various matrix metalloproteinases play in brain disorders. We summarize and review current knowledge and understanding of matrix metalloproteinases in the brain and at the blood-brain barrier in neuroinflammation, multiple sclerosis, cerebral aneurysms, stroke, epilepsy, Alzheimer's disease, Parkinson's disease, and brain cancer. We discuss the detrimental effects matrix metalloproteinases can have in these conditions, contributing to blood-brain barrier leakage, neuroinflammation, neurotoxicity, demyelination, tumor angiogenesis, and cancer metastasis. We also discuss the beneficial role matrix metalloproteinases can play in neuroprotection and anti-inflammation. Finally, we address matrix metalloproteinases as potential therapeutic targets. Together, in this comprehensive review, we summarize current understanding and knowledge of matrix metalloproteinases in the brain and at the blood-brain barrier in brain disorders.
Keywords: Alzheimer’s; Blood–brain barrier; Parkinson’s disease; epilepsy; inflammation; intracranial aneurysm; multiple sclerosis; neurodegeneration; stroke.
© The Author(s) 2016.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

