R-spondin 2 promotes acetylcholine receptor clustering at the neuromuscular junction via Lgr5
- PMID: 27328992
- PMCID: PMC4916433
- DOI: 10.1038/srep28512
R-spondin 2 promotes acetylcholine receptor clustering at the neuromuscular junction via Lgr5
Abstract
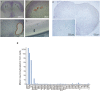
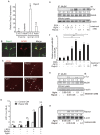
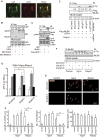
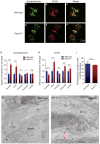
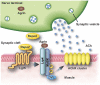
At the neuromuscular junction (NMJ), acetylcholine receptor (AChR) clustering is mediated by spinal motor neuron (SMN)-derived agrin and its receptors on the muscle, the low-density lipoprotein receptor-related protein 4 (LRP4) and muscle-specific receptor tyrosine kinase (MuSK). Additionally, AChR clustering is mediated by the components of the Wnt pathway. Laser capture microdissection of SMNs revealed that a secreted activator of Wnt signaling, R-spondin 2 (Rspo2), is highly expressed in SMNs. We found that Rspo2 is enriched at the NMJ, and that Rspo2 induces MuSK phosphorylation and AChR clustering. Rspo2 requires Wnt ligands, but not agrin, for promoting AChR clustering in cultured myotubes. Leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5), an Rspo2 receptor, is also accumulated at the NMJ, and is associated with MuSK via LRP4. Lgr5 is required for Rspo2-mediated AChR clustering in myotubes. In Rspo2-knockout mice, the number and density of AChRs at the NMJ are reduced. The Rspo2-knockout diaphragm has an altered ultrastructure with widened synaptic clefts and sparse synaptic vesicles. Frequency of miniature endplate currents is markedly reduced in Rspo2-knockout mice. To conclude, we demonstrate that Rspo2 and its receptor Lgr5 are Wnt-dependent and agrin-independent regulators of AChR clustering at the NMJ.
Figures

References
-
- Burden S. J. SnapShot: Neuromuscular Junction. Cell 144, 826–826 e821 (2011). - PubMed
-
- Sanes J. R. & Lichtman J. W. Induction, assembly, maturation and maintenance of a postsynaptic apparatus. Nat Rev Neurosci 2, 791–805 (2001). - PubMed
-
- Bezakova G. & Ruegg M. A. New insights into the roles of agrin. Nat Rev Mol Cell Biol 4, 295–308 (2003). - PubMed
-
- McMahan U. J. et al. Agrin isoforms and their role in synaptogenesis. Curr Opin Cell Biol 4, 869–874 (1992). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

