Construction and sequencing of an infectious clone of the goose embryo-adapted Muscovy duck parvovirus vaccine strain FZ91-30
- PMID: 27329377
- PMCID: PMC4915054
- DOI: 10.1186/s12985-016-0564-9
Construction and sequencing of an infectious clone of the goose embryo-adapted Muscovy duck parvovirus vaccine strain FZ91-30
Abstract
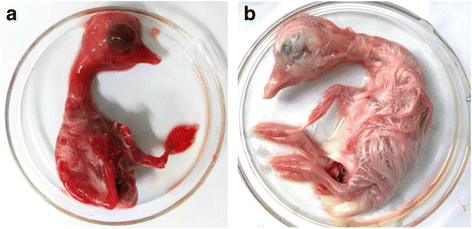
Background: Muscovy duck parvovirus (MDPV) is the etiological agent of Muscovy duckling parvoviral disease, which is characterized by diarrhea, locomotive dysfunction, stunting, and death in young ducklings, and causes substantial economic losses in the Muscovy duck industry worldwide. FZ91-30 is an attenuated vaccine strain that is safe and immunogenic to ducklings, but the genomic information and molecular mechanism underlining the attenuation are not understood.
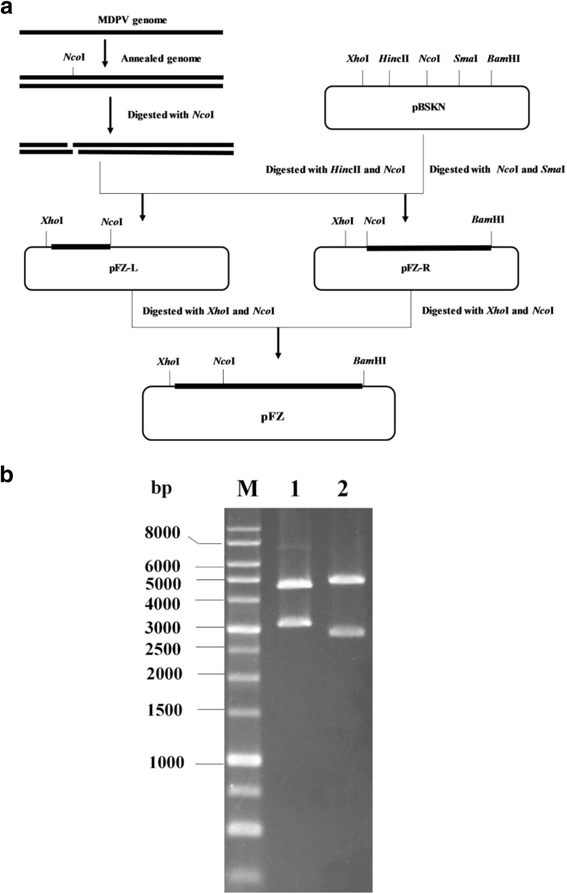
Methods: The FZ91-30 strain was propagated in 11-day-old embryonated goose eggs, and viral particles were purified from the pooled allantoic fluid by differential centrifugation and ultracentrifugation. Single-stranded genomic DNA was extracted and annealed to form double-stranded DNA. The dsDNA digested with NcoI resulted two sub-genomic fragments, which were then cloned into the modified plasmid pBluescript II SK, respectively, generating plasmid pBSKNL and pBSKNR. The sub-genomic plasmid clones were sequenced and further combined to construct the plasmid pFZ that contained the entire genome of strain FZ91-30. The complete genome sequences of strain FM and YY and partial genome sequences of other strains were retrieved from GenBank for sequence comparison. The plasmid pFZ containing the entire genome of FZ91-30 was transfected in 11-day-old embryonated goose eggs via the chorioallantoic membranes route to rescue infectious virus. A genetic marker was introduced into the rescued virus to discriminate from its parental virus.
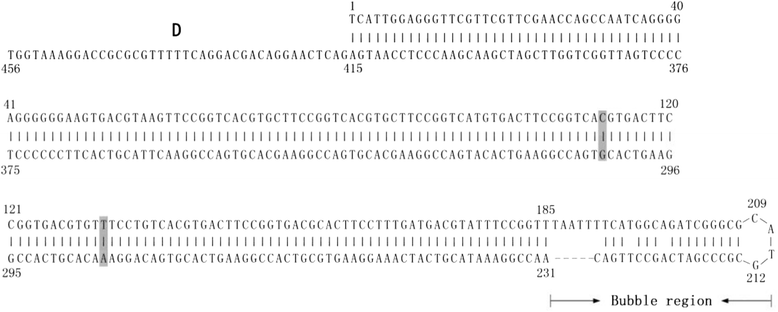
Results: The genome of FZ91-30 consists of 5,131 nucleotides and has 98.9 % similarity to the FM strain. The inverted terminal repeats (ITR) are 456 nucleotides in length, 14 nucleotides longer than that of Goose parvovirus (GPV). The exterior 415 nucleotides of the ITR form a hairpin structure, and the interior 41 nucleotides constitute the D sequence, a reverse complement of the D' sequence at the 3' ITR. Amino acid sequence alignment of the VP1 proteins between FZ91-30 and five pathogenic MDPV strains revealed that FZ91-30 had five mutations; two in the unique region of the VP1 protein (VP1u) and three in VP3. Sequence alignment of the Rep1 proteins revealed two amino acid alterations for FZ91-30, both of which were conserved for two pathogenic strains YY and P. Transfection of the plasmid pFZ in 11-day-old embryonated goose eggs resulted in generation of infectious virus with similar biological properties as compared with the parental strain.
Conclusions: The amino acid mutations identified in the VP1 and Rep1 protein may contribute to the attenuation of FZ91-30 in Muscovy ducklings. Plasmid transfection in embryonated goose eggs was suitable for rescue of infectious MDPV.
Keywords: Attenuation; Infectious clone; Muscovy duck parvovirus; Rescue; Transfection.
Figures

Similar articles
-
Molecular characterization of a novel Muscovy duck parvovirus isolate: evidence of recombination between classical MDPV and goose parvovirus strains.BMC Vet Res. 2017 Nov 9;13(1):327. doi: 10.1186/s12917-017-1238-6. BMC Vet Res. 2017. PMID: 29121936 Free PMC article.
-
Transfection of embryonated Muscovy duck eggs with a recombinant plasmid is suitable for rescue of infectious Muscovy duck parvovirus.Arch Virol. 2017 Dec;162(12):3869-3874. doi: 10.1007/s00705-017-3541-8. Epub 2017 Sep 7. Arch Virol. 2017. PMID: 28884224
-
Cloning of the genome of a goose parvovirus vaccine strain SYG61v and rescue of infectious virions from recombinant plasmid in embryonated goose eggs.J Virol Methods. 2014 May;200:41-6. doi: 10.1016/j.jviromet.2014.02.014. Epub 2014 Feb 22. J Virol Methods. 2014. PMID: 24565999
-
Avian parvovirus: classification, phylogeny, pathogenesis and diagnosis.Avian Pathol. 2018 Dec;47(6):536-545. doi: 10.1080/03079457.2018.1517938. Epub 2018 Sep 24. Avian Pathol. 2018. PMID: 30246559 Review.
-
Parvovirus variation for disease: a difference with RNA viruses?Curr Top Microbiol Immunol. 2006;299:349-70. doi: 10.1007/3-540-26397-7_13. Curr Top Microbiol Immunol. 2006. PMID: 16568906 Review.
Cited by
-
Rapid and visual detection of an isolated and identified goose parvovirus (GPV) strain by a loop-mediated isothermal amplification assay.Vet Res Forum. 2023;14(1):7-12. doi: 10.30466/vrf.2021.540351.3246. Epub 2023 Jan 15. Vet Res Forum. 2023. PMID: 36816861 Free PMC article.
-
Recombinant Muscovy Duck Parvovirus Led to Ileac Damage in Muscovy Ducklings.Viruses. 2022 Jul 3;14(7):1471. doi: 10.3390/v14071471. Viruses. 2022. PMID: 35891451 Free PMC article.
-
Development of a PCR assay for detection and differentiation of Muscovy duck and goose parvoviruses based on NS gene characterization.J Vet Med Sci. 2018 Dec 11;80(12):1861-1866. doi: 10.1292/jvms.18-0256. Epub 2018 Oct 8. J Vet Med Sci. 2018. PMID: 30298830 Free PMC article.
-
Development of a Multiplex Quantitative Polymerase Chain Reaction Assay for the Detection of Duck Enteritis Virus, Goose Parvovirus, and Muscovy Duck Parvovirus.Animals (Basel). 2025 May 29;15(11):1599. doi: 10.3390/ani15111599. Animals (Basel). 2025. PMID: 40509065 Free PMC article.
-
Identification and genomic analysis of two novel duck-origin GPV-related parvovirus in China.BMC Vet Res. 2019 Mar 12;15(1):88. doi: 10.1186/s12917-019-1833-9. BMC Vet Res. 2019. PMID: 30866923 Free PMC article.
References
-
- Jestin V, Le Bras MO, Cherbonnel M, Le Gall G, Bennejean G. Mise en évidence de parvovirus (virus de la maladie de Derzsy) très pathogènes dans les élevages de canards de Barbarie. Rec Med Vet. 1991;167:849–57.
-
- Fournier D, Gaudry D. Recent discoveries on waterfowl pathology: a new parvovirus of Muscovy ducks in France—Field vaccination trials. In: McNulty MS, McFerran JB, editors. Proceedings of the Commission of the European Communities Meeting on Virus Diseases of Poultry-New and Evolving Pathogens. Brussels: European Commission; 1992. pp. 183–94.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

