Efficacy of betamethasone valerate medicated plaster on painful chronic elbow tendinopathy: a double-blind, randomized, placebo-controlled trial
- PMID: 27331041
- PMCID: PMC4915452
- DOI: 10.11138/mltj/2016.6.1.131
Efficacy of betamethasone valerate medicated plaster on painful chronic elbow tendinopathy: a double-blind, randomized, placebo-controlled trial
Abstract
Objective: to investigate the efficacy and safety of a medicated plaster containing betamethasone valerate (BMV) 2.25 mg in patients with chronic elbow tendinopathy.
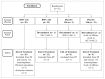
Methods: randomized, double-blind, placebo-controlled study with assignment 2:2:1:1 to BMV medicated plaster applied daily for 12 hours, daily for 24 hours or matched placebo. 62 patients aged ≥18 years with chronic lateral elbow tendinopathy were randomized. The primary efficacy variable was pain reduction (VAS) at day 28. Secondary objectives included summed pain intensity differences (SPID), overall treatment efficacy and tolerability.
Results: mean reduction in VAS pain score at day 28 was greater in both BMV medicated plaster groups, -39.35±27.69 mm for BMV12-h and -36.91±32.50 mm for BMV24-h, than with placebo, -20.20±27.32 mm. Considering the adjusted mean decreases, there was a statistically significant difference between BMV12-h and placebo (p=0.0110). Global pain relief (SPID) and overall treatment efficacy were significantly better with BMV. BMV and placebo plasters had similar local tolerability and there were few treatment-related adverse events.
Conclusions: BMV plaster was significantly more effective than placebo at reducing pain in patients with chronic elbow tendinopathies. The BMV plaster was safe and well tolerated.
Keywords: betamethasone valerate; chronic elbow tendinopathy; pain; topical corticosteroids; treatment.
Figures

Similar articles
-
Diclofenac epolamine medicated plaster in the treatment of minor soft tissue injuries: a multicenter randomized controlled trial.Curr Med Res Opin. 2013 Sep;29(9):1137-46. doi: 10.1185/03007995.2013.816669. Epub 2013 Jul 3. Curr Med Res Opin. 2013. PMID: 23777324 Clinical Trial.
-
Efficacy and safety of the Betamethasone valerate 0.1% plaster in mild-to-moderate chronic plaque psoriasis: a randomized, parallel-group, active-controlled, phase III study.Am J Clin Dermatol. 2011 Jun 1;12(3):191-201. doi: 10.2165/11539780-000000000-00000. Am J Clin Dermatol. 2011. PMID: 21284407 Clinical Trial.
-
Efficacy and Safety of Calcipotriol Plus Betamethasone Dipropionate Aerosol Foam Compared with Betamethasone 17-Valerate-Medicated Plaster for the Treatment of Psoriasis.Clin Drug Investig. 2017 Apr;37(4):355-361. doi: 10.1007/s40261-016-0489-5. Clin Drug Investig. 2017. PMID: 27995521 Free PMC article. Clinical Trial.
-
Role of betamethasone valerate 2.250 mg medicated plaster in the treatment of psoriasis and other dermatological pathologies: a review.Drugs Context. 2018 Aug 20;7:212539. doi: 10.7573/dic.212539. eCollection 2018. Drugs Context. 2018. PMID: 30151017 Free PMC article. Review.
-
The topical 5% lidocaine medicated plaster in localized neuropathic pain: a reappraisal of the clinical evidence.J Pain Res. 2016 Feb 12;9:67-79. doi: 10.2147/JPR.S99231. eCollection 2016. J Pain Res. 2016. PMID: 26929664 Free PMC article. Review.
Cited by
-
Multimodal approach to rehabilitation of the patients with lateral epicondylosis: a case series.Springerplus. 2016 Oct 5;5(1):1718. doi: 10.1186/s40064-016-3375-y. eCollection 2016. Springerplus. 2016. PMID: 27777854 Free PMC article.
-
Efficacy of transdermal anti-inflammatory patches for musculoskeletal pain: a systematic review and meta-analysis.Pain Manag. 2024 Oct-Nov;14(10-11):557-569. doi: 10.1080/17581869.2024.2421153. Epub 2024 Nov 22. Pain Manag. 2024. PMID: 39574250 Free PMC article.
-
Topical polidocanol application in combination with static stretching in tendinopathies: a prospective pilot study.Muscles Ligaments Tendons J. 2017 May 10;7(1):88-97. doi: 10.11138/mltj/2017.7.1.088. eCollection 2017 Jan-Mar. Muscles Ligaments Tendons J. 2017. PMID: 28717616 Free PMC article.
References
-
- Ahmad Z, Siddiqui N, Malik SS, Abdus-Samee M, Tytherleigh-Strong G, Rushton N. Lateral epicondylitis: a review of pathology and management. Bone Joint J. 2013;95-B(9):1158–1164. - PubMed
-
- Allander E. Prevalence, incidence and remission rates of some common rheumatic diseases or syndromes. Scand J Rheumatol. 1974;3:145–153. - PubMed
-
- Calfee RP, Patel A, DaSilva MF, Akelman E. Management of lateral epicondylitis: current concepts. J Am Acad Orthop Surg. 2008;16:19–29. - PubMed
-
- Karkhanis S, Frost A, Maffulli N. Operative management of tennis elbow: a quantitative review. Br Med Bull. 2008;88(1):171–188. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
