Adaptive Acceleration of Visually Evoked Smooth Eye Movements in Mice
- PMID: 27335412
- PMCID: PMC4916255
- DOI: 10.1523/JNEUROSCI.0067-16.2016
Adaptive Acceleration of Visually Evoked Smooth Eye Movements in Mice
Abstract
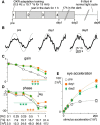
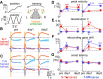
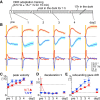
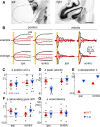
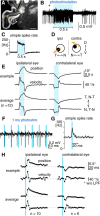
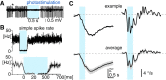
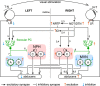
The optokinetic response (OKR) consists of smooth eye movements following global motion of the visual surround, which suppress image slip on the retina for visual acuity. The effective performance of the OKR is limited to rather slow and low-frequency visual stimuli, although it can be adaptably improved by cerebellum-dependent mechanisms. To better understand circuit mechanisms constraining OKR performance, we monitored how distinct kinematic features of the OKR change over the course of OKR adaptation, and found that eye acceleration at stimulus onset primarily limited OKR performance but could be dramatically potentiated by visual experience. Eye acceleration in the temporal-to-nasal direction depended more on the ipsilateral floccular complex of the cerebellum than did that in the nasal-to-temporal direction. Gaze-holding following the OKR was also modified in parallel with eye-acceleration potentiation. Optogenetic manipulation revealed that synchronous excitation and inhibition of floccular complex Purkinje cells could effectively accelerate eye movements in the nasotemporal and temporonasal directions, respectively. These results collectively delineate multiple motor pathways subserving distinct aspects of the OKR in mice and constrain hypotheses regarding cellular mechanisms of the cerebellum-dependent tuning of movement acceleration.
Significance statement: Although visually evoked smooth eye movements, known as the optokinetic response (OKR), have been studied in various species for decades, circuit mechanisms of oculomotor control and adaptation remain elusive. In the present study, we assessed kinematics of the mouse OKR through the course of adaptation training. Our analyses revealed that eye acceleration at visual-stimulus onset primarily limited working velocity and frequency range of the OKR, yet could be dramatically potentiated during OKR adaptation. Potentiation of eye acceleration exhibited different properties between the nasotemporal and temporonasal OKRs, indicating distinct visuomotor circuits underlying the two. Lesions and optogenetic manipulation of the cerebellum provide constraints on neural circuits mediating visually driven eye acceleration and its adaptation.
Keywords: adaptation; cerebellum; mouse; optokinetic response.
Copyright © 2016 the authors 0270-6474/16/366836-14$15.00/0.
Figures


Similar articles
-
Non-linear eye movements during visual-vestibular interaction under body oscillation with step-mode lateral linear acceleration.Exp Brain Res. 2005 Feb;161(2):243-54. doi: 10.1007/s00221-004-2063-2. Epub 2004 Oct 22. Exp Brain Res. 2005. PMID: 15502986 Clinical Trial.
-
Motor dynamics encoding in cat cerebellar flocculus middle zone during optokinetic eye movements.J Neurophysiol. 1999 Nov;82(5):2235-48. doi: 10.1152/jn.1999.82.5.2235. J Neurophysiol. 1999. PMID: 10561402
-
Comparison of plasticity and development of mouse optokinetic and vestibulo-ocular reflexes suggests differential gain control mechanisms.Vision Res. 2004 Dec;44(28):3419-27. doi: 10.1016/j.visres.2004.09.006. Vision Res. 2004. PMID: 15536010
-
LTD, RP, and Motor Learning.Cerebellum. 2016 Feb;15(1):51-53. doi: 10.1007/s12311-015-0698-0. Cerebellum. 2016. PMID: 26160222 Review.
-
Development of the optokinetic response in macaques: a comparison with cat and man.Ann N Y Acad Sci. 2003 Oct;1004:10-8. doi: 10.1196/annals.1303.002. Ann N Y Acad Sci. 2003. PMID: 14662443 Review.
Cited by
-
Purkinje cell microzones mediate distinct kinematics of a single movement.Nat Commun. 2023 Jul 19;14(1):4358. doi: 10.1038/s41467-023-40111-5. Nat Commun. 2023. PMID: 37468512 Free PMC article.
-
Population calcium responses of Purkinje cells in the oculomotor cerebellum driven by nonvisual input.J Neurophysiol. 2021 Oct 1;126(4):1391-1402. doi: 10.1152/jn.00715.2020. Epub 2021 Aug 4. J Neurophysiol. 2021. PMID: 34346783 Free PMC article.
-
Long term visuo-vestibular mismatch in freely behaving mice differentially affects gaze stabilizing reflexes.Sci Rep. 2020 Nov 18;10(1):20018. doi: 10.1038/s41598-020-77026-w. Sci Rep. 2020. PMID: 33208812 Free PMC article.
-
PyOKR: A Semi-Automated Method for Quantifying Optokinetic Reflex Tracking Ability.J Vis Exp. 2024 Apr 12;(206):10.3791/66779. doi: 10.3791/66779. J Vis Exp. 2024. PMID: 38682904 Free PMC article.
-
Mouse Extraocular Muscles and the Musculotopic Organization of Their Innervation.Anat Rec (Hoboken). 2019 Oct;302(10):1865-1885. doi: 10.1002/ar.24141. Epub 2019 May 7. Anat Rec (Hoboken). 2019. PMID: 30993879 Free PMC article.
References
-
- Barmack NH, Pettorossi VE. Effects of unilateral lesions of the flocculus on optokinetic and vestibuloocular reflexes of the rabbit. J Neurophysiol. 1985;53:481–496. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
