MAIT cells are activated during human viral infections
- PMID: 27337592
- PMCID: PMC4931007
- DOI: 10.1038/ncomms11653
MAIT cells are activated during human viral infections
Abstract
Mucosal-associated invariant T (MAIT) cells are abundant in humans and recognize bacterial ligands. Here, we demonstrate that MAIT cells are also activated during human viral infections in vivo. MAIT cells activation was observed during infection with dengue virus, hepatitis C virus and influenza virus. This activation-driving cytokine release and Granzyme B upregulation-is TCR-independent but dependent on IL-18 in synergy with IL-12, IL-15 and/or interferon-α/β. IL-18 levels and MAIT cell activation correlate with disease severity in acute dengue infection. Furthermore, HCV treatment with interferon-α leads to specific MAIT cell activation in vivo in parallel with an enhanced therapeutic response. Moreover, TCR-independent activation of MAIT cells leads to a reduction of HCV replication in vitro mediated by IFN-γ. Together these data demonstrate MAIT cells are activated following viral infections, and suggest a potential role in both host defence and immunopathology.
Figures



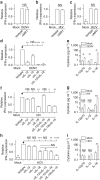
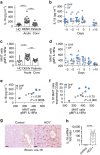
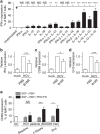

References
-
- Dusseaux M. et al.. Human MAIT cells are xenobiotic-resistant, tissue-targeted, CD161hi IL-17-secreting T cells. Blood 117, 1250–1259 (2011). - PubMed
-
- Treiner E. et al.. Selection of evolutionarily conserved mucosal-associated invariant T cells by MR1. Nature 422, 164–169 (2003). - PubMed
-
- Tang X.-Z. et al.. IL-7 licenses activation of human liver intrasinusoidal mucosal-associated invariant T cells. J. Immunol. 190, 3142–3152 (2013). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- MC_PC_12020/MRC_/Medical Research Council/United Kingdom
- MR/N012658/1/MRC_/Medical Research Council/United Kingdom
- G1002552/MRC_/Medical Research Council/United Kingdom
- MR/K010239/1/MRC_/Medical Research Council/United Kingdom
- G1100247/MRC_/Medical Research Council/United Kingdom
- U19 AI082630/AI/NIAID NIH HHS/United States
- G0600000/MRC_/Medical Research Council/United Kingdom
- G0400802/MRC_/Medical Research Council/United Kingdom
- MR/K01532X/1/MRC_/Medical Research Council/United Kingdom
- G0400720/MRC_/Medical Research Council/United Kingdom
- MC_UU_12014/9/MRC_/Medical Research Council/United Kingdom
- 109965/Z/15/Z/WT_/Wellcome Trust/United Kingdom
- G0600371/MRC_/Medical Research Council/United Kingdom
- MR/K000241/1/MRC_/Medical Research Council/United Kingdom
- 091663/WT_/Wellcome Trust/United Kingdom
- G0801508/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

