Platelet-Rich Plasma Obtained with Different Anticoagulants and Their Effect on Platelet Numbers and Mesenchymal Stromal Cells Behavior In Vitro
- PMID: 27340410
- PMCID: PMC4909912
- DOI: 10.1155/2016/7414036
Platelet-Rich Plasma Obtained with Different Anticoagulants and Their Effect on Platelet Numbers and Mesenchymal Stromal Cells Behavior In Vitro
Abstract
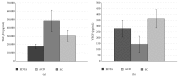
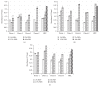
There are promising results in the use of platelet-rich plasma (PRP) for musculoskeletal tissue repair. However, the variability in the methodology for its obtaining may cause different and opposing findings in the literature. Particularly, the choice of the anticoagulant is the first definition to be made. In this work, blood was collected with sodium citrate (SC), ethylenediaminetetraacetic acid (EDTA), or anticoagulant citrate dextrose (ACD) solution A, as anticoagulants, prior to PRP obtaining. Hematological analysis and growth factors release quantification were performed, and the effects on mesenchymal stromal cell (MSC) culture, such as cytotoxicity and cell proliferation (evaluated by MTT method) and gene expression, were evaluated. The use of EDTA resulted in higher platelet yield in whole blood; however, it induced an increase in the mean platelet volume (MPV) following the blood centrifugation steps for PRP obtaining. The use of SC and ACD resulted in higher induction of MSC proliferation. On the other hand, PRP obtained in SC presented the higher platelet recovery after the blood first centrifugation step and a minimal change in MSC gene expression. Therefore, we suggest the use of SC as the anticoagulant for PRP obtaining.
Figures




References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

