Inductive Effect of Palmatine on Apoptosis in RAW 264.7 Cells
- PMID: 27340419
- PMCID: PMC4906184
- DOI: 10.1155/2016/7262054
Inductive Effect of Palmatine on Apoptosis in RAW 264.7 Cells
Abstract
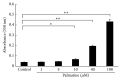
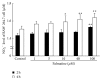
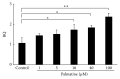
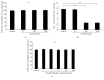
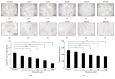
Osteoporosis is a serious public health problem characterized by low bone density and deterioration of the bone microarchitecture. Current treatment options target either osteoclast resorption or osteoblast formation. It has been reported that berberine, a close structural analog of palmatine, inhibited bone loss in an osteoporosis model. In this study, osseous metabolism was observed in vitro with osteoclast bone resorbing cells. We proved that mouse preosteoclastic cell line (RAW 264.7) has a higher sensitivity to palmatine than mouse osteoblastic cell line (MC3T3-E1); the cell survival rates significantly decreased at 40 μM palmatine. The NO2 (-) level, a metabolic product of nitric monoxide (NO), and iNOS mRNA expression, an osteoclast with NO induced enzyme, also increased with higher dosage of palmatine. Furthermore, it was recognized that the cell viability decrease from palmatine was caused by apoptosis rather than necrosis. Additionally, osteoclast apoptosis from palmatine did not occur when iNOS was inhibited with N(G)-nitro-L-arginine methyl ester hydrochloride (pan NOS inhibitor). These results indicate that palmatine plays an important role in osteoclast apoptosis via the NOS system. Hence, palmatine could be considered as a viable pharmaceutical candidate for osteoporosis bone resorption inhibitor.
Figures

Similar articles
-
Palmatine attenuates osteoclast differentiation and function through inhibition of receptor activator of nuclear factor-κb ligand expression in osteoblast cells.Biol Pharm Bull. 2010;33(10):1733-9. doi: 10.1248/bpb.33.1733. Biol Pharm Bull. 2010. PMID: 20930384
-
Influence of Palmatine on Bone Metabolism in Ovariectomized Mice and Cytokine Secretion of Osteoblasts.In Vivo. 2015 Nov-Dec;29(6):671-7. In Vivo. 2015. PMID: 26546523
-
Eldecalcitol effects on osteoblastic differentiation and function in the presence or absence of osteoclastic bone resorption.Exp Ther Med. 2019 Sep;18(3):2111-2121. doi: 10.3892/etm.2019.7784. Epub 2019 Jul 17. Exp Ther Med. 2019. PMID: 31410166 Free PMC article.
-
Strontium ranelate in osteoporosis.Curr Pharm Des. 2002;8(21):1907-16. doi: 10.2174/1381612023393639. Curr Pharm Des. 2002. PMID: 12171530 Review.
-
The action of GH/IGF-I/IGFBP in osteoblasts and osteoclasts.Horm Res. 1997;48 Suppl 5:45-9. doi: 10.1159/000191328. Horm Res. 1997. PMID: 9434044 Review.
Cited by
-
Synergistic Effect of Plant Compounds in Combination with Conventional Antimicrobials against Biofilm of Staphylococcus aureus, Pseudomonas aeruginosa, and Candida spp.Pharmaceuticals (Basel). 2023 Oct 30;16(11):1531. doi: 10.3390/ph16111531. Pharmaceuticals (Basel). 2023. PMID: 38004397 Free PMC article. Review.
-
Palmatine as an Agent Against Metabolic Syndrome and Its Related Complications: A Review.Drug Des Devel Ther. 2020 Nov 17;14:4963-4974. doi: 10.2147/DDDT.S280520. eCollection 2020. Drug Des Devel Ther. 2020. PMID: 33235437 Free PMC article. Review.
-
From traditional remedy to modern therapy: a comprehensive review of palmatine's multi-target mechanisms and ethnopharmacological potential.Front Pharmacol. 2025 Jul 25;16:1624353. doi: 10.3389/fphar.2025.1624353. eCollection 2025. Front Pharmacol. 2025. PMID: 40786032 Free PMC article. Review.
-
The herbal decoction modified Danggui Buxue Tang attenuates immune-mediated bone marrow failure by regulating the differentiation of T lymphocytes in an immune-induced aplastic anemia mouse model.PLoS One. 2017 Jul 6;12(7):e0180417. doi: 10.1371/journal.pone.0180417. eCollection 2017. PLoS One. 2017. PMID: 28683082 Free PMC article.
-
Glucagon-like peptide 2 decreases osteoclasts by stimulating apoptosis dependent on nitric oxide synthase.Cell Prolif. 2018 Aug;51(4):e12443. doi: 10.1111/cpr.12443. Epub 2018 Feb 19. Cell Prolif. 2018. PMID: 29457300 Free PMC article.
References
-
- Tamma R., Zallone A. Osteoblast and osteoclast crosstalks: from OAF to Ephrin. Inflammation & Allergy-Drug Targets. 2012;11(3):196–200. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

