Phosphoprotein Phosphatase 1 Is Required for Extracellular Calcium-Induced Keratinocyte Differentiation
- PMID: 27340655
- PMCID: PMC4909930
- DOI: 10.1155/2016/3062765
Phosphoprotein Phosphatase 1 Is Required for Extracellular Calcium-Induced Keratinocyte Differentiation
Abstract
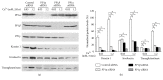
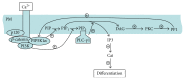
Extracellular calcium is a major regulator of keratinocyte differentiation in vitro and appears to play that role in vivo, but the mechanism is unclear. We have previously demonstrated that, following calcium stimulation, PIP5K1α is recruited by the E-cadherin-β-catenin complex to the plasma membrane where it provides the substrate PIP2 for both PI3K and PLC-γ1. This signaling pathway is critical for calcium-induced generation of second messengers including IP3 and intracellular calcium and keratinocyte differentiation. In this study, we explored the upstream regulatory mechanism by which calcium activates PIP5K1α and the role of this activation in calcium-induced keratinocyte differentiation. We found that treatment of human keratinocytes in culture with calcium resulted in an increase in serine dephosphorylation and PIP5K1α activation. PP1 knockdown blocked extracellular calcium-induced increase in serine dephosphorylation and activity of PIP5K1α and induction of keratinocyte differentiation markers. Knockdown of PLC-γ1, the downstream effector of PIP5K1α, blocked upstream dephosphorylation and PIP5K1α activation induced by calcium. Coimmunoprecipitation revealed calcium induced recruitment of PP1 to the E-cadherin-catenin-PIP5K1α complex in the plasma membrane. These results indicate that PP1 is recruited to the extracellular calcium-dependent E-cadherin-catenin-PIP5K1α complex in the plasma membrane to activate PIP5K1α, which is required for PLC-γ1 activation leading to keratinocyte differentiation.
Figures








Similar articles
-
PIP5K1α promotes myogenic differentiation via AKT activation and calcium release.Stem Cell Res Ther. 2018 Feb 9;9(1):33. doi: 10.1186/s13287-018-0770-z. Stem Cell Res Ther. 2018. PMID: 29426367 Free PMC article.
-
Phosphatidylinositol-4-phosphate 5-kinase 1alpha mediates extracellular calcium-induced keratinocyte differentiation.Mol Biol Cell. 2009 Mar;20(6):1695-704. doi: 10.1091/mbc.e08-07-0756. Epub 2009 Jan 21. Mol Biol Cell. 2009. PMID: 19158393 Free PMC article.
-
The recruitment of phosphatidylinositol 3-kinase to the E-cadherin-catenin complex at the plasma membrane is required for calcium-induced phospholipase C-gamma1 activation and human keratinocyte differentiation.J Biol Chem. 2007 Mar 23;282(12):8695-703. doi: 10.1074/jbc.M609135200. Epub 2007 Jan 22. J Biol Chem. 2007. PMID: 17242406
-
The role of phospholipase C-gamma1 in 1alpha,25-dihydroxyvitamin D(3) regulated keratinocyte differentiation.Steroids. 2001 Mar-May;66(3-5):339-45. doi: 10.1016/s0039-128x(00)00163-x. Steroids. 2001. PMID: 11179742 Review.
-
Vitamin D regulated keratinocyte differentiation: role of coactivators.J Cell Biochem. 2003 Feb 1;88(2):290-5. doi: 10.1002/jcb.10339. J Cell Biochem. 2003. PMID: 12520529 Review.
Cited by
-
PIP5K1α promotes myogenic differentiation via AKT activation and calcium release.Stem Cell Res Ther. 2018 Feb 9;9(1):33. doi: 10.1186/s13287-018-0770-z. Stem Cell Res Ther. 2018. PMID: 29426367 Free PMC article.
-
p120-catenin suppresses proliferation and tumor growth of oral squamous cell carcinoma via inhibiting nuclear phospholipase C-γ1 signaling.J Cell Physiol. 2020 Dec;235(12):9399-9413. doi: 10.1002/jcp.29744. Epub 2020 Apr 30. J Cell Physiol. 2020. PMID: 32356317 Free PMC article.
-
Various Cellular Components and Its Signaling Cascades Through the Involvement of Signaling Messengers in Keratinocyte Differentiation.Antioxidants (Basel). 2025 Apr 1;14(4):426. doi: 10.3390/antiox14040426. Antioxidants (Basel). 2025. PMID: 40298779 Free PMC article. Review.
-
HPV8-E6 Interferes with Syntenin-2 Expression through Deregulation of Differentiation, Methylation and Phosphatidylinositide-Kinase Dependent Mechanisms.Front Microbiol. 2017 Sep 8;8:1724. doi: 10.3389/fmicb.2017.01724. eCollection 2017. Front Microbiol. 2017. PMID: 28970821 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

