Involvement of human chorionic gonadotropin in regulating vasculogenic mimicry and hypoxia-inducible factor-1α expression in ovarian cancer cells
- PMID: 27346985
- PMCID: PMC4919850
- DOI: 10.1186/s12935-016-0327-0
Involvement of human chorionic gonadotropin in regulating vasculogenic mimicry and hypoxia-inducible factor-1α expression in ovarian cancer cells
Abstract
Background: Human chorionic gonadotropin (hCG) can play a crucial role in angiogenesis. In the present study, we focused on hCG to gain insight into its potential effects on vasculogenic mimicry (VM) in ovarian cancer cells.
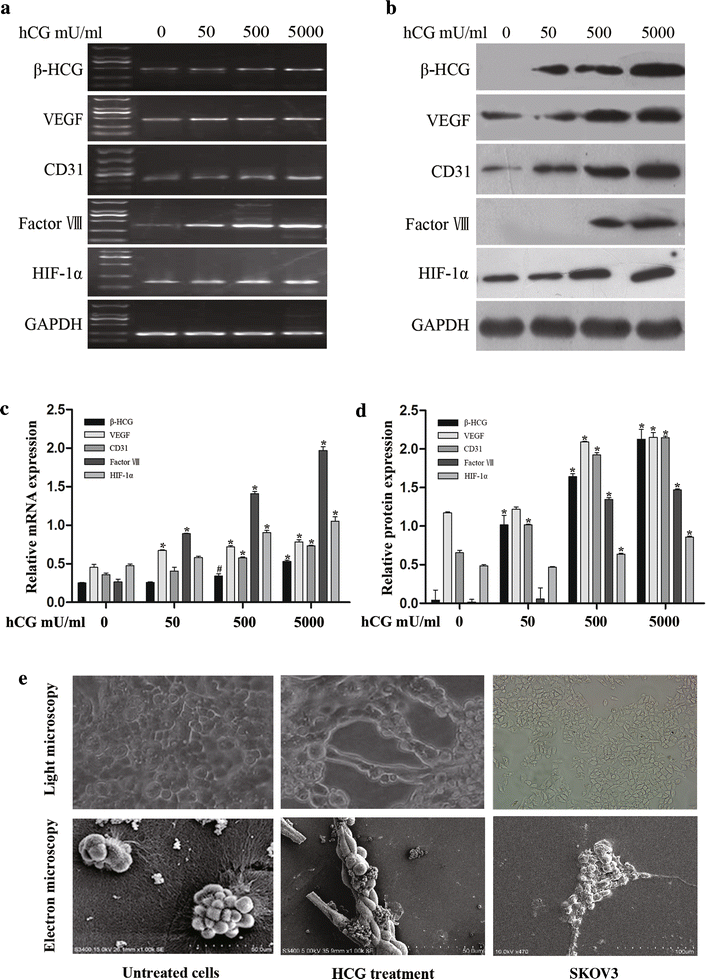
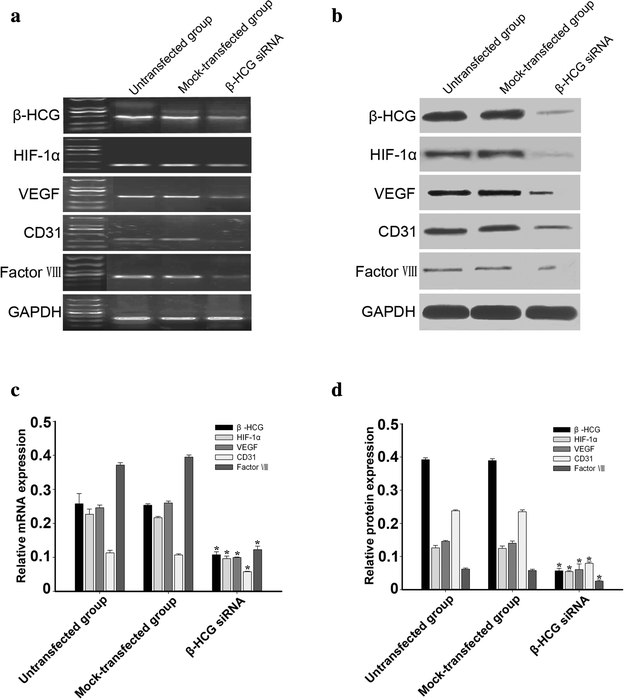
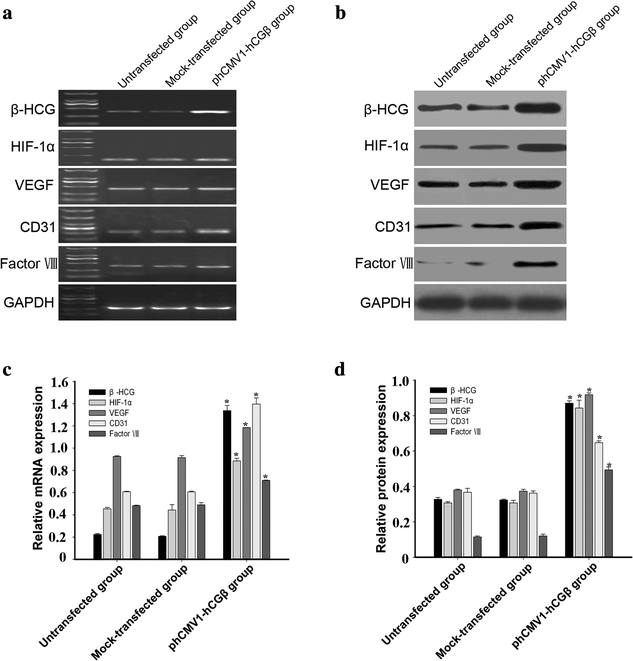
Methods: Ovarian cancer OVCAR-3 cells were incubated with different concentrations of recombinant hCG in 3-dimensional cultures. VM was identified by morphological observations and vascular endothelial cell marker detection in OVCAR-3 cells. Expression of hCG, hypoxia-inducible factor-1α (HIF-1α), and the endothelial cell markers CD31, VEGF, and factor VIII were detected by reverse transcription polymerase chain reaction and western blotting. The effect of hCG on endothelial cell-marker expression in ovarian cancer cells was further explored using small interfering RNA (siRNA) and plasmid-based approaches.
Results: Incubation of OVCAR-3 cells with recombinant hCG induced vessel-like network formation, which was accompanied by significant elevation of vascular marker expression. Attenuation of hCG expression by siRNA in OVCAR-3 cells suppressed the expression of endothelial cell markers and HIF-1α by tumour cells. Overexpression of hCG in OVCAR-3 cells resulted in increased expression of endothelial cell markers and HIF-1α.
Conclusions: HCG was crucial for changing the phenotype of OVCAR-3 cells to endothelial-like cells. The effect of hCG induction on VM in ovarian cancer cells is potentially associated with HIF-1α.
Keywords: Human chorionic gonadotropin; Hypoxia inducible factor-1α; Ovarian cancer; Vasculogenic mimicry.
Figures

References
-
- Yao LQ, Feng YJ, Ding JX, Jing HM, Xu CJ, Chen SF, et al. Differentiation of vascular endothelial cells-like derived from epithelial ovarian cancer cells induced by hypoxia. Int J Oncol. 2007;30:1069–1075. - PubMed
-
- Vartanian AA, Stepanova EV, Gutorov SL, Solomko ES, Grigorieva IN, Sokolova IN, et al. Prognostic significance of periodic acid-Schiff-positive patterns in clear cell renal cell carcinoma. Can J Urol. 2009;16:4726–4732. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

