Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) in children and adults
- PMID: 27347773
- PMCID: PMC6464789
- DOI: 10.1002/14651858.CD002787.pub3
Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) in children and adults
Abstract
Background: Acute hypoxaemic respiratory failure (AHRF) and mostly acute respiratory distress syndrome (ARDS) are critical conditions. AHRF results from several systemic conditions and is associated with high mortality and morbidity in individuals of all ages. Inhaled nitric oxide (INO) has been used to improve oxygenation, but its role remains controversial. This Cochrane review was originally published in 2003, and has been updated in 2010 and 2016.
Objectives: The primary objective was to examine the effects of administration of inhaled nitric oxide on mortality in adults and children with ARDS. Secondary objectives were to examine secondary outcomes such as pulmonary bleeding events, duration of mechanical ventilation, length of stay, etc. We conducted subgroup and sensitivity analyses, examined the role of bias and applied trial sequential analyses (TSAs) to examine the level of evidence.
Search methods: In this update, we searched the Cochrane Central Register of Controlled Trials (CENTRAL; 2015 Issue 11); MEDLINE (Ovid SP, to 18 November 2015), EMBASE (Ovid SP, to 18 November 2015), CAB, BIOSIS and the Cumulative Index to Nursing and Allied Health Literature (CINAHL). We handsearched the reference lists of the newest reviews and cross-checked them with our search of MEDLINE. We contacted the main authors of included studies to request any missed, unreported or ongoing studies. The search was run from inception until 18 November 2015.
Selection criteria: We included all randomized controlled trials (RCTs), irrespective of publication status, date of publication, blinding status, outcomes published or language. We contacted trial investigators and study authors to retrieve relevant and missing data.
Data collection and analysis: Two review authors independently extracted data and resolved disagreements by discussion. Our primary outcome measure was all-cause mortality. We performed several subgroup and sensitivity analyses to assess the effects of INO in adults and children and on various clinical and physiological outcomes. We presented pooled estimates of the effects of interventions as risk ratios (RRs) with 95% confidence intervals (CIs). We assessed risk of bias through assessment of trial methodological components and risk of random error through trial sequential analysis.
Main results: Our primary objective was to assess effects of INO on mortality. We found no statistically significant effects of INO on longest follow-up mortality: 250/654 deaths (38.2%) in the INO group compared with 221/589 deaths (37.5%) in the control group (RR 1.04, 95% CI 0.9 to 1.19; I² statistic = 0%; moderate quality of evidence). We found no statistically significant effects of INO on mortality at 28 days: 202/587 deaths (34.4%) in the INO group compared with 166/518 deaths (32.0%) in the control group (RR 1.08, 95% CI 0.92 to 1.27; I² statistic = 0%; moderate quality of evidence). In children, there was no statistically significant effects of INO on mortality: 25/89 deaths (28.1%) in the INO group compared with 34/96 deaths (35.4%) in the control group (RR 0.78, 95% CI 0.51 to 1.18; I² statistic = 22%; moderate quality of evidence).Our secondary objective was to assess the benefits and harms of INO. For partial pressure of oxygen in arterial blood (PaO2)/fraction of inspired oxygen (FiO2), we found significant improvement at 24 hours (mean difference (MD) 15.91, 95% CI 8.25 to 23.56; I² statistic = 25%; 11 trials, 614 participants; moderate quality of evidence). For the oxygenation index, we noted significant improvement at 24 hours (MD -2.31, 95% CI -2.73 to -1.89; I² statistic = 0%; five trials, 368 participants; moderate quality of evidence). For ventilator-free days, the difference was not statistically significant (MD -0.57, 95% CI -1.82 to 0.69; I² statistic = 0%; five trials, 804 participants; high quality of evidence). There was a statistically significant increase in renal failure in the INO groups (RR 1.59, 95% CI 1.17 to 2.16; I² statistic = 0%; high quality of evidence).
Authors' conclusions: Evidence is insufficient to support INO in any category of critically ill patients with AHRF. Inhaled nitric oxide results in a transient improvement in oxygenation but does not reduce mortality and may be harmful, as it seems to increase renal impairment.
Conflict of interest statement
Fabienne Gebistorf: none.
Oliver Karam: none.
Jørn Wetterslev is a member of the Copenhagen Trial Unit (CTU) task force. The CTU develop the theory and software for Trial Sequential Analysis (TSA) which is available free of charge at:
Arash Afshari: none.
Figures



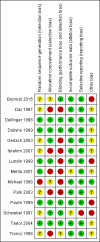




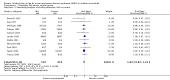

























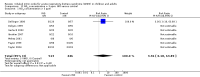
Update of
-
Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) and acute lung injury in children and adults.Cochrane Database Syst Rev. 2010 Jul 7;(7):CD002787. doi: 10.1002/14651858.CD002787.pub2. Cochrane Database Syst Rev. 2010. Update in: Cochrane Database Syst Rev. 2016 Jun 27;(6):CD002787. doi: 10.1002/14651858.CD002787.pub3. PMID: 20614430 Updated.
References
References to studies included in this review
Bronicki 2015 {published and unpublished data}
-
- Bronicki RA, Fortenberry J, Schreiber, M, Checchia PA, Anas NG. Multicenter randomized controlled trial of inhaled nitric oxide for pediatric acute respiratory distress syndrome. The Journal of Pediatrics 2015;66(2):365‐9. - PubMed
Day 1997 {published data only}
-
- Day RW, Allen EM, Witte MK. A randomized, controlled study of the 1‐hour and 24‐hour effects of inhaled nitric oxide therapy in children with acute hypoxemic respiratory failure. Chest 1997;112(5):1324‐31. [PUBMED: 9367476] - PubMed
Dellinger 1998 {published data only}
-
- Dellinger RP, Zimmerman JL, Taylor RW, Straube RC, Hauser DL, Criner GJ, et al. Effects of inhaled nitric oxide in patients with acute respiratory distress syndrome: results of a randomized phase II trial. Inhaled Nitric Oxide in ARDS Study Group. Critical Care Medicine 1998;26(1):15‐23. [PUBMED: PMID: 9428538] - PubMed
Dobyns 1999 {published data only}
-
- Dobyns EL, Anas NG, Fortenberry JD, Deshpande J, Cornfield DN, Tasker RC, et al. Interactive effects of high‐frequency oscillatory ventilation and inhaled nitric oxide in acute hypoxemic respiratory failure in pediatrics. Critical Care Medicine 2002;30(11):2425‐9. [PUBMED: 12441749] - PubMed
-
- Dobyns EL, Cornfield DN, Anas NG, Fortenberry JD, Tasker RC, Lynch A, et al. Multicenter randomized controlled trial of the effects of inhaled nitric oxide therapy on gas exchange in children with acute hypoxemic respiratory failure. The Journal of Pediatrics 1999;134(4):406‐12. [PUBMED: 10190913] - PubMed
Gerlach 2003 {published data only}
-
- Gerlach H, Keh D, Semmerow A, Busch T, Lewandowski K, Pappert DM, et al. Dose‐response characteristics during long‐term inhalation of nitric oxide in patients with severe acute respiratory distress syndrome: a prospective, randomized, controlled study. American Journal of Respiratory and Critical Care Medicine 2003;167(7):1008‐15. [PUBMED: 12663340] - PubMed
Ibrahim 2007 {published data only}
-
- Ibrahim TS, El‐Mohamady HS. Inhaled nitric oxide and prone position: how far they can improve oxygenation in pediatric patients with acute respiratory distress syndrome?. Journal of Medical Science 2007;7:390‐5.
Lundin 1999 {published data only}
-
- Lundin S, Mang H, Smithies M, Stenqvist O, Frostell C. Inhalation of nitric oxide in acute lung injury: results of a European multicentre study. The European Study Group of Inhaled Nitric Oxide. Intensive Care Medicine 1999;25(9):911‐9. [PUBMED: 10501745] - PubMed
Mehta 2001 {published data only}
-
- Mehta S, Simms HH, Levy MM, Hill NS, Schwartz W, Nelson D, et al. Inhaled nitric oxide improves oxygenation acutely but not chronically in acute respiratory distress syndrome: a randomized, controlled trial. Journal of Applied Research in Clinical and Experimental Therapeutics 2001;1:73‐84.
Michael 1998 {published data only}
-
- Michael JR, Barton RG, Saffle JR, Mone M, Markewitz BA, Hillier K, et al. Inhaled nitric oxide versus conventional therapy: effect on oxygenation in ARDS. American Journal of Respiratory and Critical Care Medicine 1998;157(5 Pt 1):1372‐80. [PUBMED: 9603111] - PubMed
Park 2003 {published data only}
-
- Park KJ, Lee YJ, Oh YJ, Lee KS, Sheen SS, Hwang SC. Combined effects of inhaled nitric oxide and a recruitment maneuver in patients with acute respiratory distress syndrome. Yonsei Medical Journal 2003;44(2):219‐26. [PUBMED: 12728461] - PubMed
Payen 1999 {published data only}
-
- Payen D, Vallet B, Group d'étude du NO dans l'ARDS. Results of the French prospective multicentric randomized double‐blind placebo‐controlled trial on inhaled nitric oxide (NO) in ARDS [abstract]. Intensive Care Medicine 1999;25 Suppl 1:166.
Schwebel 1997 {published data only}
-
- Schwebel C, Beuret P, Perdrix JP, Jospe R, Duperret S, Fogliani J, et al. Early inhaled nitric oxide inhalation in acute lung injury: results of a double‐blind randomized study [abstract]. Intensive Care Medicine 1997;23 Suppl 1:2.
Taylor 2004 {published data only}
-
- Angus DC, Clermont G, Linde‐Zwirble WT, Musthafa AA, Dremsizov TT, Lidicker J, et al. Healthcare costs and long‐term outcomes after acute respiratory distress syndrome: a phase III trial of inhaled nitric oxide. Critical Care Medicine 2006;34(12):2883‐90. [PUBMED: 17075373] - PubMed
-
- Taylor RW, Zimmerman JL, Dellinger RP, Straube RC, Criner GJ, Davis K Jr, et al. Low‐dose inhaled nitric oxide in patients with acute lung injury: a randomized controlled trial. JAMA 2004;291(13):1603‐9. [PUBMED: 15069048] - PubMed
Troncy 1998 {published data only}
-
- Troncy E, Collet JP, Shapiro S, Guimond JG, Blair L, Ducruet T, et al. Inhaled nitric oxide in acute respiratory distress syndrome: a pilot randomized controlled study. American Journal of Respiratory and Critical Care Medicine 1998;157(5 Pt 1):1483‐8. [PUBMED: 9603127] - PubMed
References to studies excluded from this review
Cuthbertson 2000 {published data only}
-
- Cuthbertson BH, Galley HF, Webster NR. Effect of inhaled nitric oxide on key mediators of the inflammatory response in patients with acute lung injury. Critical Care Medicine 2000;28:1736‐41. [PUBMED: 10890611] - PubMed
Johannigman 1997 {published data only}
-
- Johannigman JA, Davis K, Campbell RS, Luchette F, Hurst JM, Branson RD. Inhaled nitric oxide in acute respiratory distress syndrome. The Journal of Trauma 1997;43(6):904‐10. [PUBMED: 9420103] - PubMed
Khan 2009 {published data only}
-
- Khan TA, Schnickel G, Ross D, Bastani S, Laks H, Esmailian F, et al. A prospective, randomized, crossover pilot study of inhaled nitric oxide versus inhaled prostacyclin in heart transplant and lung transplant recipients. The Journal of Thoracic and Cardiovascular Surgery 2009;138(6):1417‐24. [PUBMED: 19931670] - PubMed
Meade 2003 {published data only}
-
- Meade MO, Granton JT, Matte‐Martyn A, McRae K, Weaver B, Cripps P, et al. A randomized trial of inhaled nitric oxide to prevent ischemia‐reperfusion injury after lung transplantation. American Journal of Respiratory and Critical Care Medicine 2003;167(11):1483‐9. [PUBMED: 12770854] - PubMed
Perrin 2006 {published data only}
-
- Perrin G, Roch A, Michelet P, Reynaud‐Gaubert M, Thomas P, Doddoli C, et al. Inhaled nitric oxide does not prevent pulmonary edema after lung transplantation measured by lung water content: a randomized clinical study. Chest 2006;129(4):1024‐30. [PUBMED: 16608953] - PubMed
Puybasset 1994 {published data only}
-
- Puybasset L, Rouby JJ, Mourgeon E, Stewart TE, Cluzel P, Arthoud M, et al. Inhaled nitric oxide in acute respiratory failure: dose‐response curves. Intensive Care Medicine 1994;20(5):319‐27. [PUBMED: 7930025] - PubMed
Puybasset 1995 {published data only}
-
- Puybasset L, Rouby JJ, Mourgeon E, Cluzel P, Souhil Z, Law‐Koune JD, et al. Factors influencing cardiopulmonary effects of inhaled nitric oxide in acute respiratory failure. American Journal of Respiratory and Critical Care Medicine 1995;152(1):318‐28. [PUBMED: 7599840] - PubMed
Rossaint 1995 {published data only}
-
- Rossaint R, Slama K, Gerlach H, Pappert D, Veit S, Falke K. Effects of inhaled nitric oxide on right ventricular function in severe acute respiratory distress syndrome. Intensive Care Medicine 1995;21(3):197‐203. [PUBMED: 7790604] - PubMed
References to ongoing studies
Godinez {unpublished data only}
-
- Nitric Oxide Administration for Acute Respiratory Distress Syndrome. Ongoing study October 14, 2005.
Additional references
Adhikari 2007
Altman 2003
Anderson 2003
-
- Anderson MR. Update on pediatric acute respiratory distress syndrome. Respiratory Care 2003;48(3):261‐76. [PUBMED: 12667276] - PubMed
Angus 2006
-
- Angus DC, Clermont G, Linde‐Zwirble WT, Musthafa AA, Dremsizov TT, Lidicker J, et al. Healthcare costs and long‐term outcomes after acute respiratory distress syndrome: a phase III trial of inhaled nitric oxide. Critical Care Medicine 2006;34(12):2883‐90. [PUBMED: 17075373] - PubMed
ARDS Definition Task Force 2012
-
- The ARDS Definition Task Force. Acute respiratory distress syndrome ‐ the Berlin definition. JAMA 2012;307(23):2526‐33. [PUBMED: 22797452] - PubMed
ARDS network 2000
-
- The Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. New England Journal of Medicine 2000;342(18):1301‐8. [PUBMED: 10793162] - PubMed
Barrington 2007
Beckman 1990
-
- Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA. Apparent hydroxyl radical production by peroxynitrite: implications for endothelial injury from nitric oxide and superoxide. Proceedings of the National Academy of Sciences of the United States of America 1990;87(4):1620‐4. [PUBMED: 2154753] - PMC - PubMed
Bernard 1994
-
- Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, et al. The American‐European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. American Journal of Respiratory and Critical Care Medicine 1994;149(3 Pt 1):818‐24. [PUBMED: 7509706] - PubMed
Brok 2009
-
- Brok J, Thorlund K, Wetterslev J, Gluud C. Apparently conclusive meta‐analyses may be inconclusive ‐ Trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta‐analyses. Journal of Clinical Epidemiology 2009;38(1):287‐98. [PUBMED: 18824466] - PubMed
Calfee 2007
Chan 2004
-
- Chan AW, Hróbjartsson A, Haahr MT, Gøtzsche PC, Altman DG. Empirical evidence for selective reporting of outcomes in randomized trials: comparison of protocols to published articles. JAMA 2004;291(20):2457‐65. [PUBMED: 15161896] - PubMed
Chollet‐Martin 1996
-
- Chollet‐Martin S, Gatecel C, Kermarrec N, Gougerot‐Pocidalo MA, Payen DM. Alveolar neutrophil functions and cytokine levels in patients with the adult respiratory distress syndrome during nitric oxide inhalation. American Journal of Respiratory and Critical Care Medicine 1996;153(3):985‐90. [PUBMED: 8630584] - PubMed
Dahlem 2003
-
- Dahlem P, Aalderen WM, Hamaker ME, Dijkgraaf MG, Bos AP. Incidence and short‐term outcome of acute lung injury in mechanically ventilated children. European Respiratory Journal 2003;22(6):980‐5. [PUBMED: PMID: 14680089] - PubMed
Dahlem 2007
-
- Dahlem P, Aalderen WM, Bos AP. Pediatric acute lung injury. Paediatric Respiratory Reviews 2007;8(4):348‐62. [PUBMED: 18005903] - PubMed
Dobyns 2002
-
- Dobyns EL, Anas NG, Fortenberry JD, Deshpande J, Cornfield DN, Tasker RC, et al. Interactive effects of high‐frequency oscillatory ventilation and inhaled nitric oxide in acute hypoxemic respiratory failure in pediatrics. Critical Care Medicine 2002;30(11):2425‐9. [PUBMED: 12441749] - PubMed
Fierobe 1995
-
- Fierobe L, Brunet F, Dhainaut JF, Monchi M, Belghith M, Mira JP, et al. Effect of inhaled nitric oxide on right ventricular function in adult respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine 1995;151(5):1414‐9. [PUBMED: 7735594] - PubMed
Flori 2005
-
- Flori HR, Glidden DV, Rutherford GW, Matthay MA. Pediatric acute lung injury: prospective evaluation of risk factors associated with mortality. American Journal of Respiratory and Critical Care Medicine 2005;171(9):995‐1001. [PUBMED: PMID: 15618461] - PubMed
Gotzsche 2000
Gries 1998
-
- Gries A, Bode C, Peter K, Herr A, Böhrer H, Motsch J, et al. Inhaled nitric oxide inhibits human platelet aggregation, P‐selectin expression, and fibrinogen binding in vitro and in vivo. Circulation 1998;97(15):1481‐7. [PUBMED: 9576429] - PubMed
Gries 2000
-
- Gries A, Herr A, Motsch J, Holzmann A, Weimann J, Taut F, et al. Randomized, placebo‐controlled, blinded and cross‐matched study on the antiplatelet effect of inhaled nitric oxide in healthy volunteers. Thrombosis and Haemostasis 2000;83(2):309‐15. [PUBMED: 10739391] - PubMed
Haddad 1996
-
- Haddad IY, Zhu S, Crow J, Barefield E, Gadilhe T, Matalon S. Inhibition of alveolar type II cell ATP and surfactant synthesis by nitric oxide. The American Journal of Physiology 1996;270(6 Pt 1):L898‐906. [PUBMED: 8764213] - PubMed
Higgins 2011
-
- Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Handbook for Systematic Reviews of Interventions. London: The Cochrane Collaboration, 2011. [Available from www.cochrane‐handbook.org. [DOI: 10.1002/9780470712184]]
Hsu 2008
-
- Hsu CW, Lee DL, Lin SL, Sun SF, Chang HW. The initial response to inhaled nitric oxide treatment for intensive care unit patients with acute respiratory distress syndrome. Respiration 2008;75(3):288‐95. [PUBMED: 17396026] - PubMed
Hutton 2000
-
- Hutton JL, Williamson PR. Bias in meta‐analysis due to outcome variable selection within studies. Journal of the Royal Statistical Society Series C 2000;49:359‐70.
Jain 2006
-
- Jain R, DalNogare A. Pharmacological therapy for acute respiratory distress syndrome. Mayo Clinic Proceedings 2006;81(2):205‐12. [PUBMED: 16471076] - PubMed
Kass 1998
-
- Kass LJ, Apkon M. Inhaled nitric oxide in the treatment of hypoxemic respiratory failure. Current Opinion in Pediatrics 1998;10(3):284‐90. [PUBMED: 9716891] - PubMed
Kubes 1991
Köstler 2005
-
- Köstler WJ, Rabitsch W, Locker GJ, Staudinger T, El‐Menyawi I, Frass M, et al. Influence of inhaled nitric oxide on plasma nitrate concentrations in patients with adult respiratory distress syndrome and sepsis: results of a pilot study. Journal of Clinical Anesthesia 2006;18(3):179‐84. [PUBMED: 16731319] - PubMed
Lan 1983
-
- Lan KKG, DeMets DL. Discrete sequential boundaries for clinical trials. Biometrika 1983;70:659‐63.
Li 2011
Linko 2009
-
- Linko R, Okkonen M, Pettilä V, Perttilä J, Parviainen I, Ruokonen E, et al. Acute respiratory failure in intensive care units. FINNALI: a prospective cohort study. Intensive Care Medicine 2009;35(8):1352‐61. [PUBMED: 19526218] - PubMed
Luhr 1999
-
- Luhr OR, Antonsen K, Karlsson M, Aardal S, Thorsteinsson A, Frostell CG, et al. Incidence and mortality after acute respiratory failure and acute respiratory distress syndrome in Sweden, Denmark, and Iceland. The ARF Study Group. American Journal of Respiratory and Critical Care Medicine 1999;159(6):1849‐61. [PUBMED: 10351930] - PubMed
López‐Fernández 2012
-
- López‐Fernández Y, Azagra AM, Oliva P, Modesto V, Sánchez JI, Parrilla J, et al. Pediatric acute lung injury epidemiology and natural history study: incidence and outcome of the acute respiratory distress syndrome in children. Critical Care Medicine 2012;40(12):3238‐45. [PUBMED: 22990455 ] - PubMed
MacCallum 2005
-
- MacCallum NS, Evans TW. Epidemiology of acute lung injury. Current Opinion in Critical Care 2005;11(1):43‐9. [PUBMED: 15659944] - PubMed
McAndrew 1997
-
- McAndrew J, Patel RP, Jo H, Cornwell T, Lincoln T, Moellering D, et al. The interplay of nitric oxide and peroxynitrite with signal transduction pathways: implications for disease. Seminars in Perinatology 1997;21(5):351‐66. [PUBMED: 9352609] - PubMed
Meade 2004
-
- Meade MO, Jacka MJ, Cook DJ, Dodek P, Griffith L, Guyatt GH, et al. Survey of interventions for the prevention and treatment of acute respiratory distress syndrome. Critical Care Medicine 2004;32(4):946‐54. [PUBMED: 15071383] - PubMed
Palmer 1998
-
- Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L‐arginine. Nature 1988;333(6174):664‐6. [PUBMED: 3131684] - PubMed
Petrucci 2013
Pogue 1997
-
- Pogue JM, Yusuf S. Cumulating evidence from randomized trials: utilizing sequential monitoring boundaries for cumulative meta‐analysis. Controlled Clinical Trials 1997;18(6):580‐93. [PUBMED: 9408720] - PubMed
Pogue 1998
-
- Pogue J, Yusuf S. Overcoming the limitations of current meta‐analysis of randomised controlled trials. Lancet 1998;351(9095):45‐52. [PUBMED: 9433436] - PubMed
Prodhan 2004
-
- Prodhan P, Noviski N. Pediatric acute hypoxemic respiratory failure: management of oxygenation. Journal of Intensive Care Medicine 2004;19(3):140‐53. [PUBMED: 15154995] - PubMed
Pryor 1995
-
- Pryor WA, Squadrito GL. The chemistry of peroxynitrite: a product from the reaction of nitric oxide with superoxide. The American Journal of Physiology 1995;268(5 Pt 1):L699‐722. [PUBMED: 7762673] - PubMed
RevMan 5.3.5 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan) Version 5.3.5. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2015.
Rossaint 1993
-
- Rossaint R, Falke KJ, López F, Slama K, Pison U, Zapol WM. Inhaled nitric oxide for the adult respiratory distress syndrome. New England Journal of Medicine 1993;328(6):399‐405. [PUBMED: 8357359] - PubMed
Rubenfeld 2005
-
- Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, et al. Incidence and outcomes of acute lung injury. New England Journal of Medicine 2005;353(16):1685‐93. [PUBMED: PMID: 16236739] - PubMed
Rücker 2008
-
- Rücker G, Schwarzer G, Carpenter J. Arcsine test for publication bias in meta‐analyses with binary outcomes. Statistics in Medicine 2008;27(5):746‐63. [PUBMED: 17592831] - PubMed
Sokol 2003b
-
- Sokol J, Jacobs SE, Bohn D. Inhaled nitric oxide for acute hypoxic respiratory failure in children and adults: a meta‐analysis. Anesthesia and Analgesia 2003;97(4):989‐98. [PUBMED: 14500146] - PubMed
TenHoor 2001
-
- TenHoor T, Mannino DM, Moss M. Risk factors for ARDS in the United States: analysis of the 1993 National Mortality Followback Study. Chest 2001;119(4):1179‐84. [PUBMED: 11296187] - PubMed
Thomassen 1997
-
- Thomassen MJ, Buhrow LT, Connors MJ, Kaneko FT, Erzurum SC, Kavuru MS. Nitric oxide inhibits inflammatory cytokine production by human alveolar macrophages. American Journal of Respiratory Cell and Molecular Biology 1997;17(3):279‐83. [PUBMED: 9308913 ] - PubMed
Thorlund 2009
-
- Thorlund K, Devereaux PJ, Wetterslev J, Guyatt G, Ioannidis JP, Thabane L, et al. Can trial sequential monitoring boundaries reduce spurious inferences from meta‐analyses?. International Journal of Epidemiology 2009;38(1):276‐86. [PUBMED: 18824467] - PubMed
Trial Sequential Analysis (TSA) [Computer program]
-
- Copenhagen Trial Unit, Center for Clinical Intervention Research. Trial Sequential Analysis. Copenhagen, Denmark: Copenhagen Trial Unit, Center for Clinical Intervention Research, Rigshospitalet, 2011.
Valdivielso 2002
-
- Valdivielso JM, Blantz RC. Acute renal failure: is nitric oxide the bad guy?. Antioxidants and Redox Signaling 2002;4(6):925‐34. [PUBMED: 12573141] - PubMed
Ware 2000
-
- Ware LB, Matthay MA. The acute respiratory distress syndrome. New England Journal of Medicine 2000;342(18):1334‐49. [PUBMED: 10793167] - PubMed
Wetterslev 2008
-
- Wetterslev J, Thorlund K, Brok J, Gluud C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta‐analysis. Journal of Clinical Epidemiology 2008;61(1):64‐75. [PUBMED: 18083463] - PubMed
Wetterslev 2009
Zimmerman 2009
-
- Zimmerman JJ, Akhtar SR, Caldwell E, Rubenfeld GD. Incidence and outcomes of pediatric acute lung injury. Pediatrics 2009;124(1):87‐95. [PUBMED: PMID: 19564287] - PubMed
References to other published versions of this review
Afshari 2010
Karam 2017
-
- Karam O, Gebistorf F, Wetterslev J, Afshari A. The effect of inhaled nitric oxide in acute respiratory distress syndrome in children and adults: a Cochrane Systematic Review with trial sequential analysis. Anaesthesia 2017;72(1):106‐17. [doi: 10.1111/anae.13628. Epub 2016 Oct 20] - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

