Gut microbiota and colorectal cancer
- PMID: 27350830
- PMCID: PMC4917993
- DOI: 10.1186/s41021-016-0038-8
Gut microbiota and colorectal cancer
Abstract
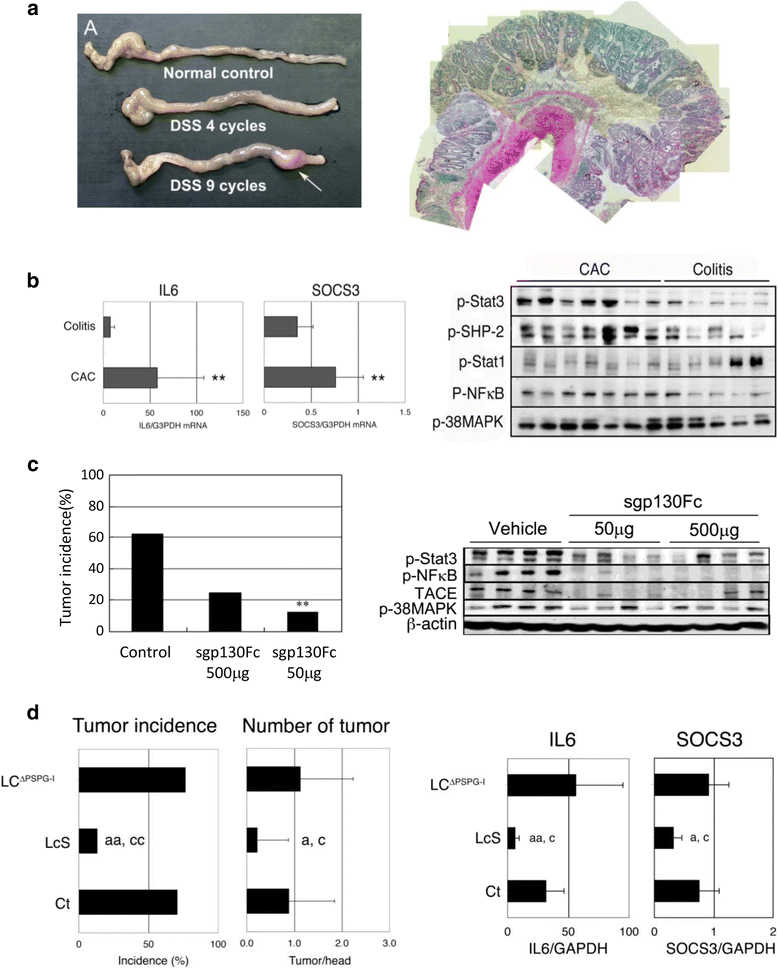
The mucosal immune system is unique to the gastrointestinal mucosa, in which a large number of immune cells are located and exert multiple functions. Meanwhile, ~100 trillion microorganisms are thought to co-inhabit in the gastrointestinal tract. Furthermore, immune cells and gut microbiota have a mutual influence and the maintenance of this symbiotic relationship results in gut homeostasis. A recent study suggested that a disturbance of gut microbiota-so called "dysbiosis"-is related to various diseases, such as inflammatory bowel disease (IBD) and colitis-associated cancer (CAC). In this review, we discuss the relationship between gut microbiota and the mucosal immune system with regard to the development of IBD and CAC. In addition, we elucidate the possibility of probiotics in treatment against these diseases.
Keywords: Colitis-associated cancer; Colorectal cancer; Dysbiosis; Gut microbiota; Mucosal immune system.
Figures

Similar articles
-
The Roles of Inflammation, Nutrient Availability and the Commensal Microbiota in Enteric Pathogen Infection.Microbiol Spectr. 2015 Jun;3(3). doi: 10.1128/microbiolspec.MBP-0008-2014. Microbiol Spectr. 2015. PMID: 26185088
-
Microbiome and colorectal cancer: Unraveling host-microbiota interactions in colitis-associated colorectal cancer development.Semin Immunol. 2017 Aug;32:3-13. doi: 10.1016/j.smim.2017.04.003. Epub 2017 May 2. Semin Immunol. 2017. PMID: 28465070 Review.
-
Mucosa-associated microbiota dysbiosis in colitis associated cancer.Gut Microbes. 2018 Mar 4;9(2):131-142. doi: 10.1080/19490976.2017.1379637. Epub 2017 Oct 12. Gut Microbes. 2018. PMID: 28914591 Free PMC article.
-
Dismicrobism in inflammatory bowel disease and colorectal cancer: changes in response of colocytes.World J Gastroenterol. 2014 Dec 28;20(48):18121-30. doi: 10.3748/wjg.v20.i48.18121. World J Gastroenterol. 2014. PMID: 25561781 Free PMC article. Review.
-
Nutrition, oxidative stress and intestinal dysbiosis: Influence of diet on gut microbiota in inflammatory bowel diseases.Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2016 Dec;160(4):461-466. doi: 10.5507/bp.2016.052. Epub 2016 Oct 26. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2016. PMID: 27812084 Review.
Cited by
-
Lacticaseibacillus rhamnosus LS8 Ameliorates Azoxymethane/Dextran Sulfate Sodium-Induced Colitis-Associated Tumorigenesis in Mice via Regulating Gut Microbiota and Inhibiting Inflammation.Probiotics Antimicrob Proteins. 2022 Oct;14(5):947-959. doi: 10.1007/s12602-022-09967-9. Epub 2022 Jul 5. Probiotics Antimicrob Proteins. 2022. PMID: 35788907
-
Gut microbiota in pathophysiology, diagnosis, and therapeutics of inflammatory bowel disease.Intest Res. 2024 Jan;22(1):15-43. doi: 10.5217/ir.2023.00080. Epub 2023 Nov 8. Intest Res. 2024. PMID: 37935653 Free PMC article. Review.
-
Commensal microbiota-derived metabolite agmatine triggers inflammation to promote colorectal tumorigenesis.Gut Microbes. 2024 Jan-Dec;16(1):2348441. doi: 10.1080/19490976.2024.2348441. Epub 2024 May 5. Gut Microbes. 2024. PMID: 38706224 Free PMC article.
-
Gut Microbiota in Colorectal Cancer: Biological Role and Therapeutic Opportunities.Cancers (Basel). 2023 Jan 30;15(3):866. doi: 10.3390/cancers15030866. Cancers (Basel). 2023. PMID: 36765824 Free PMC article. Review.
-
Increased Abundance of Clostridium and Fusobacterium in Gastric Microbiota of Patients with Gastric Cancer in Taiwan.Sci Rep. 2018 Jan 9;8(1):158. doi: 10.1038/s41598-017-18596-0. Sci Rep. 2018. PMID: 29317709 Free PMC article.
References
-
- van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, Visser CE, Kuijper EJ, Bartelsman JF, Tijssen JG, Speelman P, Dijkgraaf MG, Keller JJ. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368(5):407–15. doi: 10.1056/NEJMoa1205037. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

