Hook Adaptors Induce Unidirectional Processive Motility by Enhancing the Dynein-Dynactin Interaction
- PMID: 27365401
- PMCID: PMC5000072
- DOI: 10.1074/jbc.M116.738211
Hook Adaptors Induce Unidirectional Processive Motility by Enhancing the Dynein-Dynactin Interaction
Abstract
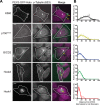
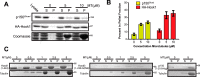
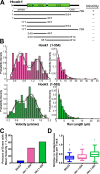
Cytoplasmic dynein drives the majority of minus end-directed vesicular and organelle motility in the cell. However, it remains unclear how dynein is spatially and temporally regulated given the variety of cargo that must be properly localized to maintain cellular function. Recent work has suggested that adaptor proteins provide a mechanism for cargo-specific regulation of motors. Of particular interest, studies in fungal systems have implicated Hook proteins in the regulation of microtubule motors. Here we investigate the role of mammalian Hook proteins, Hook1 and Hook3, as potential motor adaptors. We used optogenetic approaches to specifically recruit Hook proteins to organelles and observed rapid transport of peroxisomes to the perinuclear region of the cell. This rapid and efficient translocation of peroxisomes to microtubule minus ends indicates that mammalian Hook proteins activate dynein rather than kinesin motors. Biochemical studies indicate that Hook proteins interact with both dynein and dynactin, stabilizing the formation of a supramolecular complex. Complex formation requires the N-terminal domain of Hook proteins, which resembles the calponin-homology domain of end-binding (EB) proteins but cannot bind directly to microtubules. Single-molecule motility assays using total internal reflection fluorescence microscopy indicate that both Hook1 and Hook3 effectively activate cytoplasmic dynein, inducing longer run lengths and higher velocities than the previously characterized dynein activator bicaudal D2 (BICD2). Together, these results suggest that dynein adaptors can differentially regulate dynein to allow for organelle-specific tuning of the motor for precise intracellular trafficking.
Keywords: cytoskeleton; dynein; intracellular trafficking; kinesin; microtubule.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures





References
-
- van Spronsen M., Mikhaylova M., Lipka J., Schlager M. A., van den Heuvel D. J., Kuijpers M., Wulf P. S., Keijzer N., Demmers J., Kapitein L. C., Jaarsma D., Gerritsen H. C., Akhmanova A., and Hoogenraad C. C. (2013) TRAK/Milton motor-adaptor proteins steer mitochondrial trafficking to axons and dendrites. Neuron 77, 485–502 - PubMed
-
- Karki S., and Holzbaur E. L. (1995) affinity chromatography demonstrates a direct binding between cytoplasmic dynein and the dynactin complex. J. Biol. Chem. 270, 28806–28811 - PubMed
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

