Inflammatory related gene IKKα, IKKβ, IKKγ cooperates to determine liver cancer stem cells progression by altering telomere via heterochromatin protein 1-HOTAIR axis
- PMID: 27367027
- PMCID: PMC5226573
- DOI: 10.18632/oncotarget.10321
Inflammatory related gene IKKα, IKKβ, IKKγ cooperates to determine liver cancer stem cells progression by altering telomere via heterochromatin protein 1-HOTAIR axis
Abstract
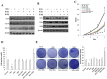
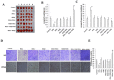
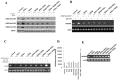
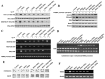
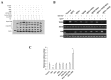
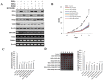
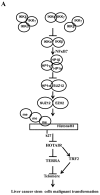
Cancer stem cells are associated with tumor recurrence. IKK is a protein kinase that is composed of IKKα, IKKβ, IKKγ. Herein, we demonstrate that IKKα plus IKKβ promoted and IKKγ inhibited liver cancer stem cell growth in vitro and in vivo. Mechanistically, IKKα plus IKKβ enhanced and IKKγ inhibited the interplay among HP1α, HP1β and HP1γ that competes for the interaction among HP1α, SUZ12, HEZ2. Therefore, IKKα plus IKKβ inhibited and IKKγ enhanced the activity of H3K27 methyltransferase SUZ12 and EZH2, which methylates H3K27 immediately sites on HOTAIR promoter region. Therefore, IKKα plus IKKβ increased and IKKγ decreased the HOTAIR expression. Strikingly, IKKα plus IKKβ decreases and IKKγ increases the HP1α interplays with DNA methyltransferase DNMT3b, which increases or decreases TERRA promoter DNA methylation. Thus IKKα plus IKKβ reduces and IKKγ increases to recruit TRF1 and RNA polymerase II deposition and elongation on the TERRA promoter locus, which increases or decreases TERRA expression. Furthermore, IKKα plus IKKβ decreases/increases and IKKγ increases/decreases the interplay between TERT and TRRRA/between TERT and TREC. Ultimately, IKKα plus IKKβ increases and IKKγ decreases the telomerase activity. On the other hand, at the telomere locus, IKKα plus IKKβ increases/drcreases and IKKγ decreases/increases TRF2, POT1, pPOT1, Exo1, pExo1, SNM1B, pSNM1B/CST-AAF binding, which keep active telomere regulatory genes and poised for telomere length. Strikingly, HOTAIR is required for IKKα plus IKKβ and IKKγ to control telomerase activity and telomere length. These observations suggest that HOTAIR operates the action of IKKα, IKKβ, IKKγ in liver cancer stem cells. This study provides a novel basis to elucidate the oncogenic action of IKKα, IKKβ, IKKγ and prompts that IKKα, IKKβ, IKKγ cooperate to HOTAR to be used as a novel therapeutic targets for liver cancer.
Keywords: HOTAIR; IKKα; IKKβ; IKKγ; telomere.
Conflict of interest statement
The authors disclose no conflicts.
Figures



References
-
- Huang JJ, Chu HX, Jiang ZY, Zhang XJ, Sun HP, You QD. Recent advances in the structure-based and ligand-based design of IKKβ inhibitors as anti-inflammation and anti-cancer agents. Curr Med Chem. 2014;21:3893–91. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

