Structure/function correlations over binuclear non-heme iron active sites
- PMID: 27369780
- PMCID: PMC5010389
- DOI: 10.1007/s00775-016-1372-9
Structure/function correlations over binuclear non-heme iron active sites
Abstract
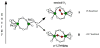
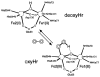
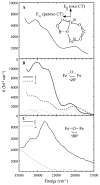
Binuclear non-heme iron enzymes activate O2 to perform diverse chemistries. Three different structural mechanisms of O2 binding to a coupled binuclear iron site have been identified utilizing variable-temperature, variable-field magnetic circular dichroism spectroscopy (VTVH MCD). For the μ-OH-bridged Fe(II)2 site in hemerythrin, O2 binds terminally to a five-coordinate Fe(II) center as hydroperoxide with the proton deriving from the μ-OH bridge and the second electron transferring through the resulting μ-oxo superexchange pathway from the second coordinatively saturated Fe(II) center in a proton-coupled electron transfer process. For carboxylate-only-bridged Fe(II)2 sites, O2 binding as a bridged peroxide requires both Fe(II) centers to be coordinatively unsaturated and has good frontier orbital overlap with the two orthogonal O2 π* orbitals to form peroxo-bridged Fe(III)2 intermediates. Alternatively, carboxylate-only-bridged Fe(II)2 sites with only a single open coordination position on an Fe(II) enable the one-electron formation of Fe(III)-O2 (-) or Fe(III)-NO(-) species. Finally, for the peroxo-bridged Fe(III)2 intermediates, further activation is necessary for their reactivities in one-electron reduction and electrophilic aromatic substitution, and a strategy consistent with existing spectral data is discussed.
Keywords: Binuclear non-heme iron enzymes; Frontier molecular orbitals; O2 activation; Peroxide activation; Variable-temperature, variable-field magnetic circular dichroism.
Figures



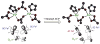


Similar articles
-
Spectroscopic and computational studies of the de novo designed protein DF2t: correlation to the biferrous active site of ribonucleotide reductase and factors that affect O2 reactivity.J Am Chem Soc. 2005 Nov 23;127(46):16098-106. doi: 10.1021/ja053661a. J Am Chem Soc. 2005. PMID: 16287296
-
Geometric and electronic structure studies of the binuclear nonheme ferrous active site of toluene-4-monooxygenase: parallels with methane monooxygenase and insight into the role of the effector proteins in O2 activation.J Am Chem Soc. 2008 Jun 4;130(22):7098-109. doi: 10.1021/ja800654d. Epub 2008 May 14. J Am Chem Soc. 2008. PMID: 18479085
-
O2 Activation by Non-Heme Iron Enzymes.Biochemistry. 2016 Nov 22;55(46):6363-6374. doi: 10.1021/acs.biochem.6b00635. Epub 2016 Nov 14. Biochemistry. 2016. PMID: 27792301 Free PMC article.
-
Diversity of structures and functions of oxo-bridged non-heme diiron proteins.Arch Biochem Biophys. 2021 Jul 15;705:108917. doi: 10.1016/j.abb.2021.108917. Epub 2021 May 12. Arch Biochem Biophys. 2021. PMID: 33991497 Free PMC article. Review.
-
Spectroscopy and electronic structures of mono- and binuclear high-valent non-heme iron-oxo systems.J Inorg Biochem. 2006 Apr;100(4):697-706. doi: 10.1016/j.jinorgbio.2006.01.013. Epub 2006 Feb 28. J Inorg Biochem. 2006. PMID: 16510189 Review.
Cited by
-
Diiron monooxygenases in natural product biosynthesis.Nat Prod Rep. 2018 Jul 18;35(7):646-659. doi: 10.1039/C7NP00061H. Nat Prod Rep. 2018. PMID: 29552683 Free PMC article. Review.
-
Generation of a μ-1,2-hydroperoxo FeIIIFeIII and a μ-1,2-peroxo FeIVFeIII Complex.Nat Commun. 2022 Mar 16;13(1):1376. doi: 10.1038/s41467-022-28894-5. Nat Commun. 2022. PMID: 35296656 Free PMC article.
-
Spectroscopy and DFT Calculations of a Flavo-diiron Enzyme Implicate New Diiron Site Structures.J Am Chem Soc. 2017 Aug 30;139(34):12009-12019. doi: 10.1021/jacs.7b06546. Epub 2017 Aug 16. J Am Chem Soc. 2017. PMID: 28756660 Free PMC article.
-
In-crystal reaction cycle of a toluene-bound diiron hydroxylase.Nature. 2017 Apr 13;544(7649):191-195. doi: 10.1038/nature21681. Epub 2017 Mar 27. Nature. 2017. PMID: 28346937
-
De Novo Design of Four-Helix Bundle Metalloproteins: One Scaffold, Diverse Reactivities.Acc Chem Res. 2019 May 21;52(5):1148-1159. doi: 10.1021/acs.accounts.8b00674. Epub 2019 Apr 11. Acc Chem Res. 2019. PMID: 30973707 Free PMC article.
References
-
- Solomon EI, Brunold TC, Davis MI, Kemsley JN, Lee SK, Lehnert N, Neese F, Skulan AJ, Yang YS, Zhou J. Chem Rev. 2000;100:235–350. - PubMed
-
- Ziehl M, He J, Dahse H-M, Hertweck C. Angew Chem Int Ed Engl. 2005;44:1202–1205. - PubMed
-
- Yang B, Hodgkinson A, Millward BA, Demaine AG. Int J Diabetes Mellit. 2010;2:169–174.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

