HIV-Exposed Uninfected Infants in Zimbabwe: Insights into Health Outcomes in the Pre-Antiretroviral Therapy Era
- PMID: 27375613
- PMCID: PMC4893498
- DOI: 10.3389/fimmu.2016.00190
HIV-Exposed Uninfected Infants in Zimbabwe: Insights into Health Outcomes in the Pre-Antiretroviral Therapy Era
Abstract
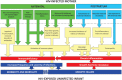
The ZVITAMBO trial recruited 14,110 mother-infant pairs to a randomized controlled trial of vitamin A between 1997 and 2000, before the availability of antiretroviral therapy for HIV prophylaxis or treatment in Zimbabwe. The HIV status of mothers and infants was well characterized through 1-2 years of follow-up, leading to the largest cohort to date of HIV-exposed uninfected (HEU) infants (n = 3135), with a suitable comparison group of HIV-unexposed infants (n = 9510). Here, we draw on 10 years of published findings from the ZVITAMBO trial. HEU infants had increased morbidity compared to HIV-unexposed infants, with 50% more hospitalizations in the neonatal period and 30% more sick clinic visits during infancy, particularly for skin infections, lower respiratory tract infections, and oral thrush. HEU children had 3.9-fold and 2.0-fold higher mortality than HIV-unexposed children during the first and second years of life, respectively, most commonly due to acute respiratory infections, diarrhea/dysentery, malnutrition, sepsis, and meningitis. Infant morbidity and mortality were strongly related to maternal HIV disease severity, and increased morbidity remained until maternal CD4 counts were >800 cells/μL. HEU infants were more likely to be premature and small-for-gestational age than HIV-unexposed infants, and had more postnatal growth failure. Here, we propose a conceptual framework to explain the increased risk of infectious morbidity, mortality, and growth failure among HEU infants, hypothesizing that immune activation and inflammation are key drivers of both infection susceptibility and growth failure. Future studies should further dissect the causes of infection susceptibility and growth failure and determine the impact of ART and cotrimoxazole on outcomes of this vulnerable group of infants in the current era.
Keywords: Africa; HIV exposure; Zimbabwe; breast-feeding; immune activation; infant; inflammation.
Figures
References
-
- Humphrey JH, Iliff PJ, Marinda ET, Mutasa K, Moulton LH, Chidawanyika H, et al. Effects of a single large dose of vitamin A, given during the postpartum period to HIV-positive women and their infants, on child HIV infection, HIV-free survival, and mortality. J Infect Dis (2006) 193(6):860–71.10.1086/500366 - DOI - PubMed
-
- Malaba LC, Iliff PJ, Nathoo KJ, Marinda E, Moulton LH, Zijenah LS, et al. Effect of postpartum maternal or neonatal vitamin A supplementation on infant mortality among infants born to HIV-negative mothers in Zimbabwe. Am J Clin Nutr (2005) 81(2):454–60. - PubMed
-
- Koyanagi A, Humphrey JH, Ntozini R, Nathoo K, Moulton LH, Iliff P, et al. Morbidity among human immunodeficiency virus-exposed but uninfected, human immunodeficiency virus-infected, and human immunodeficiency virus-unexposed infants in Zimbabwe before availability of highly active antiretroviral therapy. Pediatr Infect Dis J (2011) 30(1):45–51.10.1097/INF.0b013e3181ecbf7e - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

