Unique Transcompartmental Bridge: Antigen-Presenting Cells Sampling across Endothelial and Mucosal Barriers
- PMID: 27375624
- PMCID: PMC4901051
- DOI: 10.3389/fimmu.2016.00231
Unique Transcompartmental Bridge: Antigen-Presenting Cells Sampling across Endothelial and Mucosal Barriers
Abstract
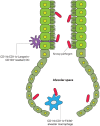
Potentially harmful pathogens can gain access to tissues and organ systems through body sites that are in direct contact with the outside environment, such as the skin, the gut, and the airway mucosa. Antigen-presenting cells (APCs) represent a bridge between the innate and adaptive immunity, and their capacity for constant immune surveillance and rapid sampling of incoming pathogens and other potentially harmful antigens is central for mounting an effective and robust protective host response. The classical view is that APCs perform this task efficiently within the tissue to sense invading agents intra-compartmentally. However, recent data based on high resolution imaging support an additional transcompartmental surveillance behavior by APC by reaching across intact physical barriers. In this review, we summarize intravital microscopic evidences of APC to sample antigens transcompartmentally at the gut mucosa and other body sites.
Keywords: T cell activation; antigen capture; antigen presentation; antigen-presenting cell; cell migration; dendritic cell; mucosal immunity; transcompartment.
Figures




References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

