Unique CD8+ T Cell-Mediated Immune Responses Primed in the Liver
- PMID: 27379551
- PMCID: PMC4935926
- DOI: 10.1097/TP.0000000000001290
Unique CD8+ T Cell-Mediated Immune Responses Primed in the Liver
Abstract
Background: The liver immune environment is tightly regulated to balance immune activation with immune tolerance. Understanding the dominant immune pathways initiated in the liver is important because the liver is a site for cell transplantation, such as for islet and hepatocyte transplantation. The purpose of this study is to examine the consequences of alloimmune stimulation when allogeneic cells are transplanted to the liver in comparison to a different immune locale, such as the kidney.
Methods: We investigated cellular and humoral immune responses when allogeneic hepatocytes are transplanted directly to the recipient liver by intraportal injection. A heterotopic kidney engraftment site was used for comparison to immune activation in the liver microenvironment.
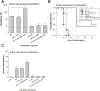
Results: Transplantation of allogeneic hepatocytes delivered directly to the liver, via recipient portal circulation, stimulated long-term, high magnitude CD8 T cell-mediated allocytotoxicity. CD8 T cells initiated significant in vivo allocytotoxicity as well as rapid rejection of hepatocytes transplanted to the liver even in the absence of secondary lymph nodes or CD4 T cells. In contrast, in the absence of recipient peripheral lymphoid tissue and CD4 T cells, CD8-mediated in vivo allocytotoxicity was abrogated, and rejection was delayed when hepatocellular allografts were transplanted to the kidney subcapsular site.
Conclusions: These results highlight the CD8-dominant proinflammatory immune responses unique to the liver microenvironment. Allogeneic cells transplanted directly to the liver do not enjoy immune privilege but rather require immunosuppression to prevent rejection by a robust and persistent CD8-dependent allocytotoxicity primed in the liver.
Conflict of interest statement
The authors of this manuscript have no conflicts of interest to disclose.
Figures



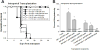
References
-
- Asakura H, Ku G, Kataoka M, Flye MW. Regulatory cells develop after the spontaneous acceptance of rat liver allografts. Surgery. 2004;136(3):532–536. - PubMed
-
- Li W, Kuhr CS, Zheng XX, et al. New insights into mechanisms of spontaneous liver transplant tolerance: the role of Foxp3-expressing CD25+CD4+ regulatory T cells. Am J Transplant. 2008;8(8):1639–1651. - PubMed
-
- Wang C, Sun J, Li L, Wang L, Dolan P, Sheil AG. Conversion of pancreas allograft rejection to acceptance by liver transplantation. Transplantation. 1998;65(2):188–192. - PubMed
-
- Meyer D, Thorwarth WM, Otto C, et al. Orthotopic liver/small bowel transplantation in rats: a microsurgical model inducing tolerance. Microsurgery. 2001;21(4):156–162. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

