Continued Risk of Relapse Independent of Treatment Modality in Limited-Stage Diffuse Large B-Cell Lymphoma: Final and Long-Term Analysis of Southwest Oncology Group Study S8736
- PMID: 27382104
- PMCID: PMC5012710
- DOI: 10.1200/JCO.2015.65.4582
Continued Risk of Relapse Independent of Treatment Modality in Limited-Stage Diffuse Large B-Cell Lymphoma: Final and Long-Term Analysis of Southwest Oncology Group Study S8736
Abstract
Purpose: Utility of combined-modality therapy for patients with limited-stage diffuse large B-cell lymphoma (DLBCL) was shown in the Southwest Oncology Group (SWOG) S8736 study, where three cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) plus radiotherapy (CHOP3RT) improved 5-year progression-free (PFS) and overall survival (OS) compared with eight cycles of CHOP (CHOP8). Subsequent analysis showed an unexpected overlap of the PFS curves. We aimed to confirm and investigate this observation by performing long-term analysis of SWOG S8736 and evaluating these data alongside data from similar patients receiving rituximab and CHOP3RT (SWOG S0014 study).
Patients and methods: A subset of patients with limited-stage DLBCL randomly assigned to CHOP8 (n = 150) or CHOP3RT (n = 158) in S8736 was analyzed along with a 56-patient subset treated in S0014 for long-term PFS and OS.
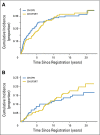
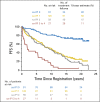
Results: Median follow-up in S8736 was 17.7 years. In patients receiving CHOP8 and CHOP3RT, median PFS was 12.0 (95% CI, 8.8 to 14.3) and 11.1 years (95% CI, 8.9 to 14.4), respectively. There were no statistically significant differences in PFS between the groups (P = .73). Median OS was 13.0 (95% CI, 10.4 to 15.2) and 13.7 years (95% CI, 11.1 to 19.4) for patients treated with CHOP8 and CHOP3RT, respectively. Similarly, there were no statistically significant differences in OS between the groups (P = .38). With a median follow-up time 12 years in S0014, 5- and 10-year OS were 82% and 67%, respectively, with a persistent pattern of relapse despite the addition of rituximab.
Conclusion: Although 5-year PFS and OS were improved after early analysis in patients with limited-stage DLBCL receiving CHOP3RT versus CHOP8, extended survival data showed similar PFS and OS, with continuous treatment failure. The addition of rituximab (S0014) to combined-modality therapy did not mitigate the continued relapse risk, underscoring the value of prolonged clinical trial patient observation and possible unique biology of limited-stage DLBCL.
Trial registration: ClinicalTrials.gov NCT00005089.
© 2016 by American Society of Clinical Oncology.
Conflict of interest statement
Authors’ disclosures of potential conflicts of interest are found in the article online at www.jco.org. Author contributions are found at the end of this article.
Figures



References
-
- Coiffier B, Thieblemont C, Van Den Neste E, et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: A study by the Groupe d’Etudes des Lymphomes de l’Adulte. Blood. 2010;116:2040–2045. - PMC - PubMed
-
- Lister TA, Crowther D, Sutcliffe SB, et al. Report of a committee convened to discuss the evaluation and staging of patients with Hodgkin’s disease: Cotswolds meeting. J Clin Oncol. 1989;7:1630–1636. - PubMed
-
- Swerdlow SH, Campo E, Harris NL, et al. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissue. (ed 4) Lyon, France: International Agency on Research for Cancer; 2008.
-
- Miller TP, Dahlberg S, Cassady JR, et al. Chemotherapy alone compared with chemotherapy plus radiotherapy for localized intermediate- and high-grade non-Hodgkin’s lymphoma. N Engl J Med. 1998;339:21–26. - PubMed
-
- Pfreundschuh M, Trümper L, Osterborg A, et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: A randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol. 2006;7:379–391. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Associated data
Grants and funding
- U10 CA180834/CA/NCI NIH HHS/United States
- UG1 CA233178/CA/NCI NIH HHS/United States
- U10 CA180835/CA/NCI NIH HHS/United States
- U10 CA180801/CA/NCI NIH HHS/United States
- UG1 CA189853/CA/NCI NIH HHS/United States
- UG1 CA189808/CA/NCI NIH HHS/United States
- UG1 CA189957/CA/NCI NIH HHS/United States
- U10 CA180888/CA/NCI NIH HHS/United States
- U10 CA180830/CA/NCI NIH HHS/United States
- UG1 CA189822/CA/NCI NIH HHS/United States
- UG1 CA189952/CA/NCI NIH HHS/United States
- U10 CA180846/CA/NCI NIH HHS/United States
- UG1 CA180830/CA/NCI NIH HHS/United States
- UG1 CA189860/CA/NCI NIH HHS/United States
- UG1 CA189830/CA/NCI NIH HHS/United States
- UG1 CA189954/CA/NCI NIH HHS/United States
- U10 CA180818/CA/NCI NIH HHS/United States
- U10 CA180828/CA/NCI NIH HHS/United States
- U10 CA180819/CA/NCI NIH HHS/United States
- UG1 CA189953/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

