Caenorhabditis elegans employs innate and learned aversion in response to bacterial toxic metabolites tambjamine and violacein
- PMID: 27384057
- PMCID: PMC4935850
- DOI: 10.1038/srep29284
Caenorhabditis elegans employs innate and learned aversion in response to bacterial toxic metabolites tambjamine and violacein
Abstract
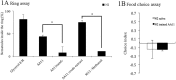
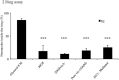
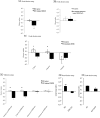
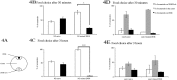
Bacteriovorus eukaryotes such as nematodes are one of the major natural predators of bacteria. In their defense bacteria have evolved a number of strategies to avoid predation, including the production of deterrent or toxic metabolites, however little is known regarding the response of predators towards such bacterial defenses. Here we use the nematode C. elegans as a model to study a predators' behavioral response towards two toxic bacterial metabolites, tambjamine YP1 and violacein. We found that C. elegans displays an innate avoidance behavior towards tambjamine YP1, however requires previous exposure to violacein before learning to avoid this metabolite. The learned avoidance of violacein is specific, reversible, is mediated via the nematode olfactory apparatus (aversive olfactory learning) and is reduced in the absence of the neurotransmitter serotonin. These multiple strategies to evade bacterial toxic metabolites represent a valuable behavioral adaptation allowing bacteriovorus predators to distinguish between good and bad food sources, thus contributing to the understanding of microbial predator-prey interactions.
Figures
Similar articles
-
A natural odor attraction between lactic acid bacteria and the nematode Caenorhabditis elegans.ISME J. 2016 Mar;10(3):558-67. doi: 10.1038/ismej.2015.134. Epub 2015 Aug 4. ISME J. 2016. PMID: 26241504 Free PMC article.
-
Pathogenic bacteria induce aversive olfactory learning in Caenorhabditis elegans.Nature. 2005 Nov 10;438(7065):179-84. doi: 10.1038/nature04216. Nature. 2005. PMID: 16281027
-
Host-microbe interactions and the behavior of Caenorhabditis elegans.J Neurogenet. 2020 Sep-Dec;34(3-4):500-509. doi: 10.1080/01677063.2020.1802724. Epub 2020 Aug 12. J Neurogenet. 2020. PMID: 32781873 Free PMC article. Review.
-
Intraguild predation between Pristionchus pacificus and Caenorhabditis elegans: a complex interaction with the potential for aggressive behaviour.J Neurogenet. 2020 Sep-Dec;34(3-4):404-419. doi: 10.1080/01677063.2020.1833004. Epub 2020 Oct 15. J Neurogenet. 2020. PMID: 33054476 Free PMC article. Review.
-
Functional organization of a neural network for aversive olfactory learning in Caenorhabditis elegans.Neuron. 2010 Dec 22;68(6):1173-86. doi: 10.1016/j.neuron.2010.11.025. Neuron. 2010. PMID: 21172617 Free PMC article.
Cited by
-
The Bacterial Gq Signal Transduction Inhibitor FR900359 Impairs Soil-Associated Nematodes.J Chem Ecol. 2023 Oct;49(9-10):549-569. doi: 10.1007/s10886-023-01442-1. Epub 2023 Jul 15. J Chem Ecol. 2023. PMID: 37453001 Free PMC article.
-
The structure and function of pathogen disgust.Philos Trans R Soc Lond B Biol Sci. 2018 Jul 19;373(1751):20170208. doi: 10.1098/rstb.2017.0208. Philos Trans R Soc Lond B Biol Sci. 2018. PMID: 29866921 Free PMC article.
-
An oxytocin-dependent social interaction between larvae and adult C. elegans.Sci Rep. 2017 Aug 31;7(1):10122. doi: 10.1038/s41598-017-09350-7. Sci Rep. 2017. PMID: 28860630 Free PMC article.
-
Multi-target drug with potential applications: violacein in the spotlight.World J Microbiol Biotechnol. 2021 Aug 16;37(9):151. doi: 10.1007/s11274-021-03120-4. World J Microbiol Biotechnol. 2021. PMID: 34398340 Review.
-
Making "Sense" of Ecology from a Genetic Perspective: Caenorhabditis elegans, Microbes and Behavior.Metabolites. 2022 Nov 9;12(11):1084. doi: 10.3390/metabo12111084. Metabolites. 2022. PMID: 36355167 Free PMC article. Review.
References
-
- Pernthaler J. Predation on prokaryotes in the water column and its ecological implications. Nat Rev Micro 3, 537–546 (2005). - PubMed
-
- Matz C. & Kjelleberg S. Off the hook - How bacteria survive protozoan grazing. Trend Microbiol 13, 302–307 (2005). - PubMed
-
- Koh K. S. et al. Minimal increase in genetic diversity enhances predation resistance. Mol Ecol 21, 1741–1753 (2012). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

