Analysis of phosphatases in ER-negative breast cancers identifies DUSP4 as a critical regulator of growth and invasion
- PMID: 27393618
- PMCID: PMC4963453
- DOI: 10.1007/s10549-016-3892-y
Analysis of phosphatases in ER-negative breast cancers identifies DUSP4 as a critical regulator of growth and invasion
Abstract
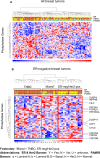
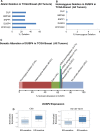
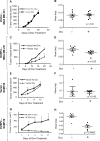
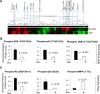
Estrogen receptor (ER)-negative cancers have a poor prognosis, and few targeted therapies are available for their treatment. Our previous analyses have identified potential kinase targets critical for the growth of ER-negative, progesterone receptor (PR)-negative and HER2-negative, or "triple-negative" breast cancer (TNBC). Because phosphatases regulate the function of kinase signaling pathways, in this study, we investigated whether phosphatases are also differentially expressed in ER-negative compared to those in ER-positive breast cancers. We compared RNA expression in 98 human breast cancers (56 ER-positive and 42 ER-negative) to identify phosphatases differentially expressed in ER-negative compared to those in ER-positive breast cancers. We then examined the effects of one selected phosphatase, dual specificity phosphatase 4 (DUSP4), on proliferation, cell growth, migration and invasion, and on signaling pathways using protein microarray analyses of 172 proteins, including phosphoproteins. We identified 48 phosphatase genes are significantly differentially expressed in ER-negative compared to those in ER-positive breast tumors. We discovered that 31 phosphatases were more highly expressed, while 11 were underexpressed specifically in ER-negative breast cancers. The DUSP4 gene is underexpressed in ER-negative breast cancer and is deleted in approximately 50 % of breast cancers. Induced DUSP4 expression suppresses both in vitro and in vivo growths of breast cancer cells. Our studies show that induced DUSP4 expression blocks the cell cycle at the G1/S checkpoint; inhibits ERK1/2, p38, JNK1, RB, and NFkB p65 phosphorylation; and inhibits invasiveness of TNBC cells. These results suggest that that DUSP4 is a critical regulator of the growth and invasion of triple-negative breast cancer cells.
Keywords: MAPK pathways; Mouse xenograft; Phosphatase; TNBC.
Figures


References
-
- Dowsett M, Cuzick J, Ingle J, Coates A, Forbes J, Bliss J, Buyse M, Baum M, Buzdar A, Colleoni M, Coombes C, Snowdon C, Gnant M, Jakesz R, Kaufmann M, Boccardo F, Godwin J, Davies C, Peto R. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28(3):509–518. doi: 10.1200/JCO.2009.23.1274. - DOI - PubMed
-
- Jakesz R, Jonat W, Gnant M, Mittlboeck M, Greil R, Tausch C, Hilfrich J, Kwasny W, Menzel C, Samonigg H, Seifert M, Gademann G, Kaufmann M, Wolfgang J. Switching of postmenopausal women with endocrine-responsive early breast cancer to anastrozole after 2 years’ adjuvant tamoxifen: combined results of ABCSG trial 8 and ARNO 95 trial. Lancet. 2005;366(9484):455–462. doi: 10.1016/S0140-6736(05)67059-6. - DOI - PubMed
-
- Mouridsen H, Giobbie-Hurder A, Goldhirsch A, Thurlimann B, Paridaens R, Smith I, Mauriac L, Forbes J, Price KN, Regan MM, Gelber RD, Coates AS. Letrozole therapy alone or in sequence with tamoxifen in women with breast cancer. N Engl J Med. 2009;361(8):766–776. doi: 10.1056/NEJMoa0810818. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

