A novel FOXM1 isoform, FOXM1D, promotes epithelial-mesenchymal transition and metastasis through ROCKs activation in colorectal cancer
- PMID: 27399334
- PMCID: PMC5311249
- DOI: 10.1038/onc.2016.249
A novel FOXM1 isoform, FOXM1D, promotes epithelial-mesenchymal transition and metastasis through ROCKs activation in colorectal cancer
Abstract
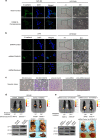
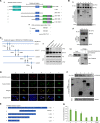
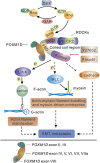
Epithelial-mesenchymal transition (EMT) is a critical event in metastasis of colorectal cancer (CRC). Rho/ROCKs signaling has a pivotal role in orchestrating actin cytoskeleton, leading to EMT and cancer invasion. However, the underlying mechanisms for ROCKs activation are not fully understood. Here, we identified FOXM1D, a novel isoform of Forkhead box M1 (FOXM1) that has a pivotal role in ROCKs activation by directly interacting with coiled-coil region of ROCK2. FOXM1D overexpression significantly polymerizes actin assembly and impairs E-cadherin expression, resulting in EMT and metastasis in xenograft mouse model and knockdown of FOXM1D has the opposite effect. Moreover, a high FOXM1D level correlates closely with clinical CRC metastasis. FOXM1D-induced ROCKs activation could be abrogated by the ROCKs inhibitors Y-27632 and fasudil. These observations indicate that the FOXM1D-ROCK2 interaction is crucial for Rho/ROCKs signaling and provide novel insight into actin cytoskeleton regulation and therapeutic potential for CRC metastasis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin 2014; 64: 9–29. - PubMed
-
- Liu X, Ji Q, Fan Z, Li Q. Cellular signaling pathways implicated in metastasis of colorectal cancer and the associated targeted agents. Future Oncol 2015; 11: 2911–2922. - PubMed
-
- Chaffer CL, Weinberg RA. A perspective on cancer cell metastasis. Science 2011; 331: 1559–1564. - PubMed
-
- Etienne-Manneville S, Hall A. Rho GTPases in cell biology. Nature 2002; 420: 629–635. - PubMed
-
- Riento K, Ridley AJ. Rocks: multifunctional kinases in cell behaviour. Nat Rev Mol Cell Biol 2003; 4: 446–456. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

