Microfluidic blood-brain barrier model provides in vivo-like barrier properties for drug permeability screening
- PMID: 27399645
- PMCID: PMC6650146
- DOI: 10.1002/bit.26045
Microfluidic blood-brain barrier model provides in vivo-like barrier properties for drug permeability screening
Abstract
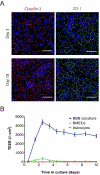
Efficient delivery of therapeutics across the neuroprotective blood-brain barrier (BBB) remains a formidable challenge for central nervous system drug development. High-fidelity in vitro models of the BBB could facilitate effective early screening of drug candidates targeting the brain. In this study, we developed a microfluidic BBB model that is capable of mimicking in vivo BBB characteristics for a prolonged period and allows for reliable in vitro drug permeability studies under recirculating perfusion. We derived brain microvascular endothelial cells (BMECs) from human induced pluripotent stem cells (hiPSCs) and cocultured them with rat primary astrocytes on the two sides of a porous membrane on a pumpless microfluidic platform for up to 10 days. The microfluidic system was designed based on the blood residence time in human brain tissues, allowing for medium recirculation at physiologically relevant perfusion rates with no pumps or external tubing, meanwhile minimizing wall shear stress to test whether shear stress is required for in vivo-like barrier properties in a microfluidic BBB model. This BBB-on-a-chip model achieved significant barrier integrity as evident by continuous tight junction formation and in vivo-like values of trans-endothelial electrical resistance (TEER). The TEER levels peaked above 4000 Ω · cm2 on day 3 on chip and were sustained above 2000 Ω · cm2 up to 10 days, which are the highest sustained TEER values reported in a microfluidic model. We evaluated the capacity of our microfluidic BBB model to be used for drug permeability studies using large molecules (FITC-dextrans) and model drugs (caffeine, cimetidine, and doxorubicin). Our analyses demonstrated that the permeability coefficients measured using our model were comparable to in vivo values. Our BBB-on-a-chip model closely mimics physiological BBB barrier functions and will be a valuable tool for screening of drug candidates. The residence time-based design of a microfluidic platform will enable integration with other organ modules to simulate multi-organ interactions on drug response. Biotechnol. Bioeng. 2017;114: 184-194. © 2016 Wiley Periodicals, Inc.
Keywords: TEER; blood-brain barrier; human iPS cells; organ on a chip; permeability.
© 2016 Wiley Periodicals, Inc.
Figures


References
-
- Abaci HE, Gledhill K, Guo Z, Christiano AM, Shuler ML. 2015. Pumpless microfluidic platform for drug testing on human skin equivalents. Lab Chip 15:882–888. http://xlink.rsc.org/?DOI=C4LC00999A. - PMC - PubMed
-
- Abaci H, Shuler M. 2015. Human-on-a-Chip Design Strategies and Principles for Physiologically Based Pharmocokinetics/Pharmacodynamics Modeling. Integr. Biol. 7:383–391. http://pubs.rsc.org/en/Content/ArticleLanding/2015/IB/C4IB00292J. - PMC - PubMed
-
- Achyuta AKH, Conway AJ, Crouse RB, Bannister EC, Lee RN, Katnik CP, Behensky A a.,Cuevas J, Sundaram SS. 2013. A modular approach to create a neurovascularunit-on-a-chip. Lab Chip 212:542–553. - PubMed
-
- Avdeef A 2012. Absorption and Drug Development: Solubility, Permeability, and Charge State. Absorpt. Drug Dev. Solubility, Permeability, Charg. State.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

