Chemokine CXCL13 mediates orofacial neuropathic pain via CXCR5/ERK pathway in the trigeminal ganglion of mice
- PMID: 27401148
- PMCID: PMC4940825
- DOI: 10.1186/s12974-016-0652-1
Chemokine CXCL13 mediates orofacial neuropathic pain via CXCR5/ERK pathway in the trigeminal ganglion of mice
Abstract
Background: Trigeminal nerve damage-induced neuropathic pain is a severely debilitating chronic orofacial pain syndrome. Spinal chemokine CXCL13 and its receptor CXCR5 were recently demonstrated to play a pivotal role in the pathogenesis of spinal nerve ligation-induced neuropathic pain. Whether and how CXCL13/CXCR5 in the trigeminal ganglion (TG) mediates orofacial pain are unknown.
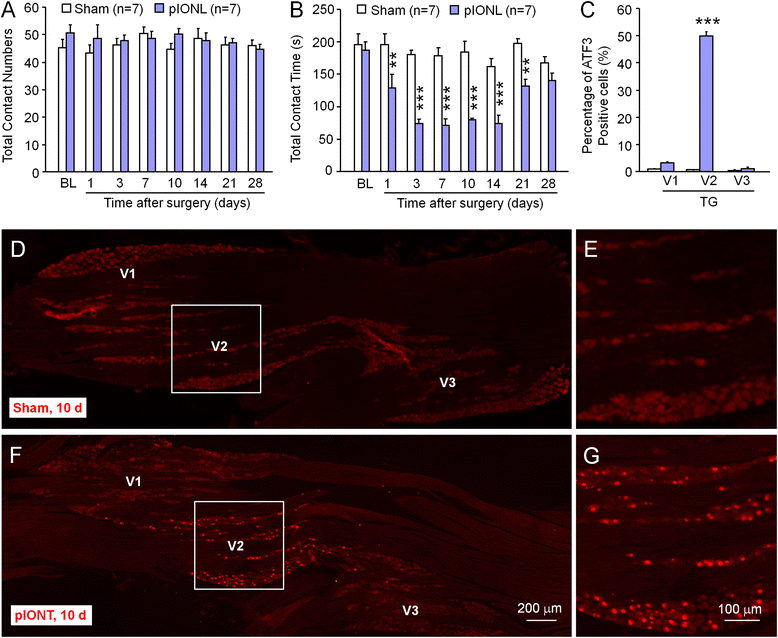
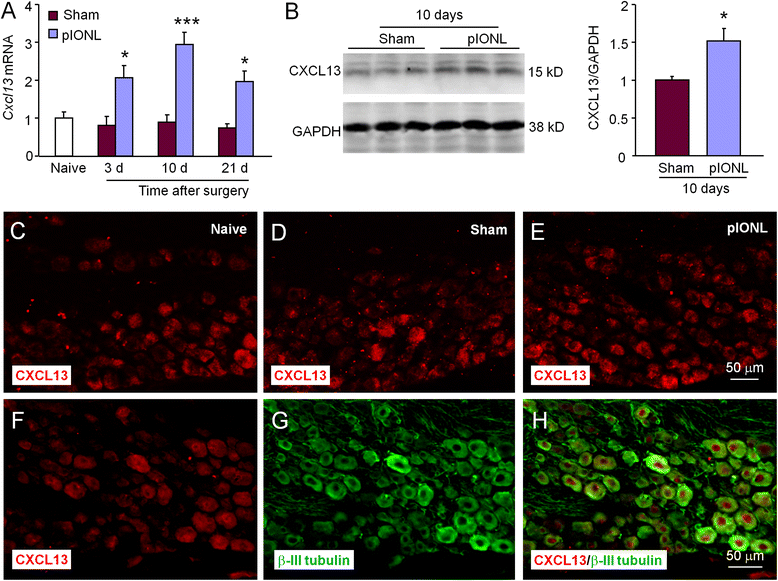
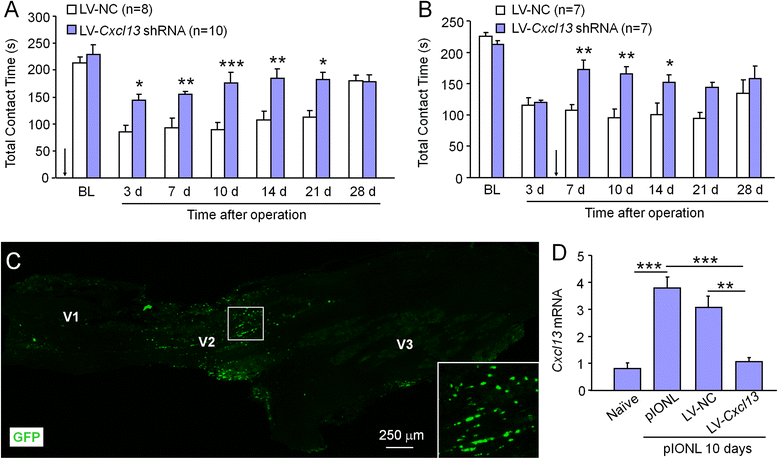
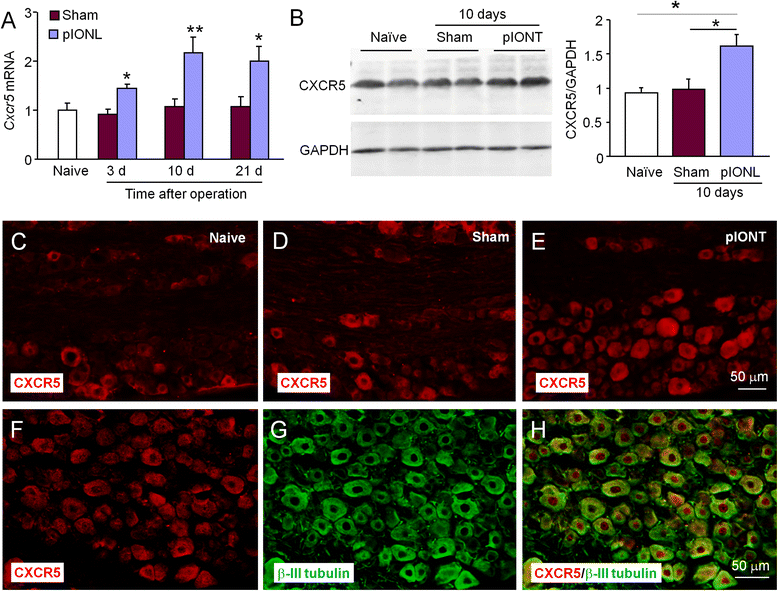
Methods: The partial infraorbital nerve ligation (pIONL) was used to induce trigeminal neuropathic pain in mice. The expression of ATF3, CXCL13, CXCR5, and phosphorylated extracellular signal-regulated kinase (pERK) in the TG was detected by immunofluorescence staining and western blot. The effect of shRNA targeting on CXCL13 or CXCR5 on pain hypersensitivity was checked by behavioral testing.
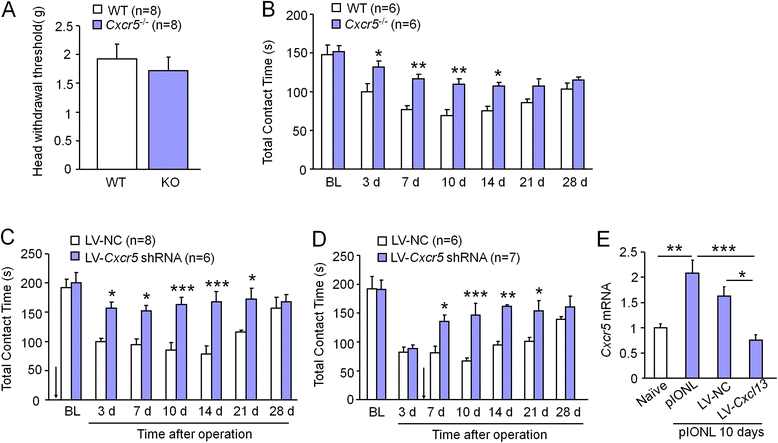
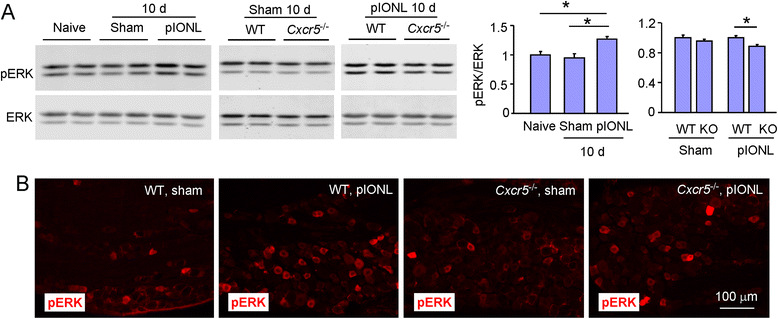
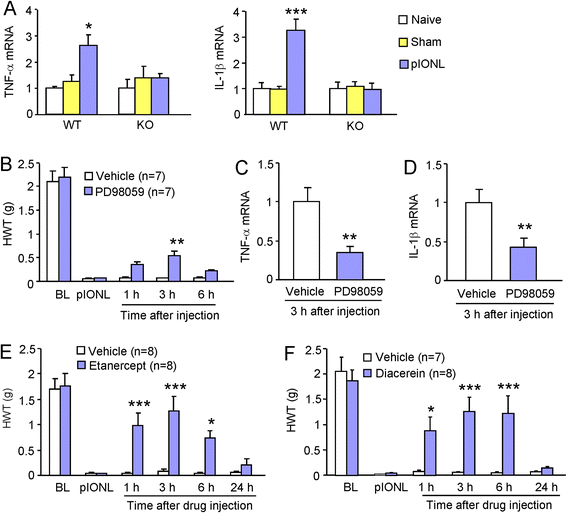
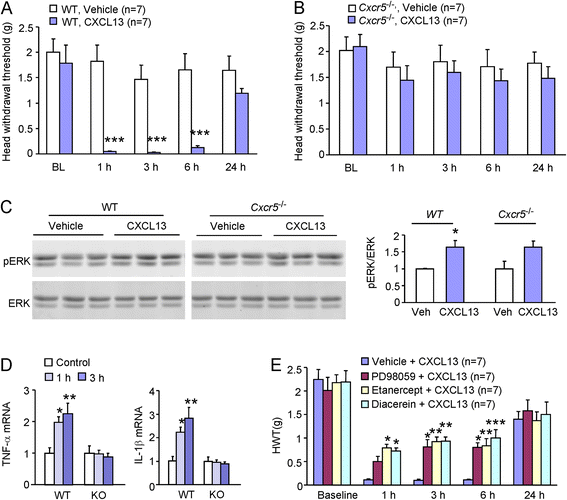
Results: pIONL induced persistent mechanical allodynia and increased the expression of ATF3, CXCL13, and CXCR5 in the TG. Inhibition of CXCL13 or CXCR5 by shRNA lentivirus attenuated pIONL-induced mechanical allodynia. Additionally, pIONL-induced neuropathic pain and the activation of ERK in the TG were reduced in Cxcr5 (-/-) mice. Furthermore, MEK inhibitor (PD98059) attenuated mechanical allodynia and reduced TNF-α and IL-1β upregulation induced by pIONL. TNF-α inhibitor (Etanercept) and IL-1β inhibitor (Diacerein) attenuated pIONL-induced orofacial pain. Finally, intra-TG injection of CXCL13 induced mechanical allodynia, increased the activation of ERK and the production of TNF-α and IL-1β in the TG of WT mice, but not in Cxcr5 (-/-) mice. Pretreatment with PD98059, Etanercept, or Diacerein partially blocked CXCL13-induced mechanical allodynia, and PD98059 also reduced CXCL13-induced TNF-α and IL-1β upregulation.
Conclusions: CXCL13 and CXCR5 contribute to orofacial pain via ERK-mediated proinflammatory cytokines production. Targeting CXCL13/CXCR5/ERK/TNF-α and IL-1β pathway in the trigeminal ganglion may offer effective treatment for orofacial neuropathic pain.
Keywords: CXCL13; CXCR5; ERK; Orofacial pain; Proinflammatory cytokines; Trigeminal ganglion.
Figures
Similar articles
-
Chemokine CXCL13 activates p38 MAPK in the trigeminal ganglion after infraorbital nerve injury.Inflammation. 2017 Jun;40(3):762-769. doi: 10.1007/s10753-017-0520-x. Inflammation. 2017. PMID: 28155010
-
CXCL13/CXCR5 signaling contributes to diabetes-induced tactile allodynia via activating pERK, pSTAT3, pAKT pathways and pro-inflammatory cytokines production in the spinal cord of male mice.Brain Behav Immun. 2019 Aug;80:711-724. doi: 10.1016/j.bbi.2019.05.020. Epub 2019 May 14. Brain Behav Immun. 2019. PMID: 31100371
-
Chemokine CCL7 mediates trigeminal neuropathic pain via CCR2/CCR3-ERK pathway in the trigeminal ganglion of mice.Mol Pain. 2023 Jan-Dec;19:17448069231169373. doi: 10.1177/17448069231169373. Mol Pain. 2023. PMID: 36998150 Free PMC article.
-
Non-neuronal cells act as crucial players in neuropathic orofacial pain.J Oral Biosci. 2024 Sep;66(3):491-495. doi: 10.1016/j.job.2024.07.005. Epub 2024 Jul 18. J Oral Biosci. 2024. PMID: 39032826 Review.
-
Cytoarchitecture and intercellular interactions in the trigeminal ganglion: Associations with neuropathic pain in the orofacial region.J Oral Biosci. 2024 Sep;66(3):485-490. doi: 10.1016/j.job.2024.07.003. Epub 2024 Jul 18. J Oral Biosci. 2024. PMID: 39032827 Review.
Cited by
-
Assessment of Growth Factors, Cytokines, and Cellular Markers in Saliva of Patients with Trigeminal Neuralgia.Molecules. 2021 May 17;26(10):2964. doi: 10.3390/molecules26102964. Molecules. 2021. PMID: 34067581 Free PMC article.
-
Pathophysiology of Post-Traumatic Trigeminal Neuropathic Pain.Biomolecules. 2022 Nov 25;12(12):1753. doi: 10.3390/biom12121753. Biomolecules. 2022. PMID: 36551181 Free PMC article. Review.
-
Peripheral CCL2-CCR2 signalling contributes to chronic headache-related sensitization.Brain. 2023 Oct 3;146(10):4274-4291. doi: 10.1093/brain/awad191. Brain. 2023. PMID: 37284790 Free PMC article.
-
An Investigation of the Molecular Mechanisms Underlying the Analgesic Effect of Jakyak-Gamcho Decoction: A Network Pharmacology Study.Evid Based Complement Alternat Med. 2020 Dec 1;2020:6628641. doi: 10.1155/2020/6628641. eCollection 2020. Evid Based Complement Alternat Med. 2020. PMID: 33343676 Free PMC article.
-
CXCL13 expressed on inflamed cerebral blood vessels recruit IL-21 producing TFH cells to damage neurons following stroke.J Neuroinflammation. 2022 May 27;19(1):125. doi: 10.1186/s12974-022-02490-2. J Neuroinflammation. 2022. PMID: 35624463 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

