Survival Outcomes of Concurrent Treatment with Docetaxel and Androgen Deprivation Therapy in Metastatic Castration-Resistant Prostate Cancer
- PMID: 27401636
- PMCID: PMC4960371
- DOI: 10.3349/ymj.2016.57.5.1070
Survival Outcomes of Concurrent Treatment with Docetaxel and Androgen Deprivation Therapy in Metastatic Castration-Resistant Prostate Cancer
Abstract
Purpose: Docetaxel-based chemotherapy (DTX) improves overall survival (OS) of men with metastatic castration-resistant prostate cancer (mCRPC). Considering the potential existence of androgen receptors that remain active at this stage, we aimed to assess the impact of the combined use of androgen deprivation therapy (ADT) with DTX for mCRPC.
Materials and methods: We performed a single-institutional retrospective analysis of patients with mCRPC who received either DTX alone (DTX group, n=21) or concurrent DTX and ADT (DTX+ADT group, n=26) between August 2006 and February 2014. All patients received DTX doses of 75 mg/m² every three weeks for at least three cycles. In the DTX+ADT group, all patients used luteinizing hormone releasing hormone agonist continuously as a concurrent ADT.
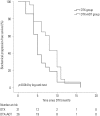
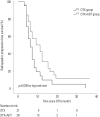
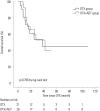
Results: The median follow-up period was 24.0 months (interquartile range 12.0-37.0) for the entire cohort. The median radiographic progression-free survival (rPFS) was 9.0 months and 6.0 months in the DTX+ADT and DTX groups, respectively (log-rank p=0.036). On multivariable Cox regression analysis, concurrent administration of ADT was the only significant predictor of rPFS [hazard ratio (HR)=0.525, 95% confidence intervals (CI) 0.284-0.970, p=0.040]. The median OS was 42.0 and 38.0 months in the DTX+ADT and DTX groups, respectively (log-rank p=0.796). On multivariable analysis, hemoglobin level at the time of DTX initiation was associated with OS (HR=0.532, 95% CI 0.381-0.744, p<0.001).
Conclusion: In chemotherapy-naive patients with mCRPC, the combined use of ADT with DTX improved rPFS. Our result suggests that the concurrent administration of ADT and DTX is superior to DTX alone.
Keywords: Prostatic neoplasms, castration-resistant; disease-free survival; docetaxel; drug therapy, combination; gonadotropin-releasing hormone; neoplasm metastasis.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures
Similar articles
-
Anticancer Activity and Tolerance of Treatments Received Beyond Progression in Men Treated Upfront with Androgen Deprivation Therapy With or Without Docetaxel for Metastatic Castration-naïve Prostate Cancer in the GETUG-AFU 15 Phase 3 Trial.Eur Urol. 2018 May;73(5):696-703. doi: 10.1016/j.eururo.2017.09.022. Epub 2017 Oct 23. Eur Urol. 2018. PMID: 29074061 Clinical Trial.
-
Prior Endocrine Therapy Impact on Abiraterone Acetate Clinical Efficacy in Metastatic Castration-resistant Prostate Cancer: Post-hoc Analysis of Randomised Phase 3 Studies.Eur Urol. 2016 May;69(5):924-32. doi: 10.1016/j.eururo.2015.10.021. Epub 2015 Oct 24. Eur Urol. 2016. PMID: 26508309 Free PMC article. Clinical Trial.
-
Neutrophil-to-lymphocyte ratio as a prognostic biomarker for men with metastatic castration-resistant prostate cancer receiving first-line chemotherapy: data from two randomized phase III trials.Ann Oncol. 2015 Apr;26(4):743-749. doi: 10.1093/annonc/mdu569. Epub 2014 Dec 15. Ann Oncol. 2015. PMID: 25515657 Clinical Trial.
-
[Treatment situation in metastastic Castration Naive Prostate Cancer (mCRPC) and the implications on clinical routine].Urologe A. 2019 Sep;58(9):1066-1072. doi: 10.1007/s00120-019-0925-2. Urologe A. 2019. PMID: 31041460 Review. German.
-
Should docetaxel be standard of care for patients with metastatic hormone-sensitive prostate cancer? Pro and contra.Ann Oncol. 2015 Aug;26(8):1660-7. doi: 10.1093/annonc/mdv245. Epub 2015 May 22. Ann Oncol. 2015. PMID: 26002607 Free PMC article. Review.
Cited by
-
Unraveling the Peculiar Features of Mitochondrial Metabolism and Dynamics in Prostate Cancer.Cancers (Basel). 2023 Feb 13;15(4):1192. doi: 10.3390/cancers15041192. Cancers (Basel). 2023. PMID: 36831534 Free PMC article. Review.
-
Estimation of Prostate Cancer Cost in Egypt From a Societal Perspective.Glob J Qual Saf Healthc. 2023 Jun 13;6(2):33-41. doi: 10.36401/JQSH-22-20. eCollection 2023 May. Glob J Qual Saf Healthc. 2023. PMID: 37333760 Free PMC article.
-
Dissecting the Hormonal Signaling Landscape in Castration-Resistant Prostate Cancer.Cells. 2021 May 7;10(5):1133. doi: 10.3390/cells10051133. Cells. 2021. PMID: 34067217 Free PMC article. Review.
-
Gonadotropin-Releasing Hormone Receptors in Prostate Cancer: Molecular Aspects and Biological Functions.Int J Mol Sci. 2020 Dec 14;21(24):9511. doi: 10.3390/ijms21249511. Int J Mol Sci. 2020. PMID: 33327545 Free PMC article. Review.
-
Efficacy of Androgen Deprivation Therapy in Patients with Metastatic Castration-Resistant Prostate Cancer Receiving Docetaxel-Based Chemotherapy.World J Mens Health. 2020 Apr;38(2):226-235. doi: 10.5534/wjmh.190029. Epub 2019 Jun 4. World J Mens Health. 2020. PMID: 31190487 Free PMC article.
References
-
- GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 11 [Internet] Lyon (France): International Agency for Research on Cancer; c2013. [updated 2014]. [cited 2015 Aug 4]. Available at: http://globocan.iarc.fr.
-
- Mohler JL, Kantoff PW, Armstrong AJ, Bahnson RR, Cohen M, D'Amico AV, et al. Prostate cancer, version 2.2014. J Natl Compr Canc Netw. 2014;12:686–718. - PubMed
-
- Lam JS, Leppert JT, Vemulapalli SN, Shvarts O, Belldegrun AS. Secondary hormonal therapy for advanced prostate cancer. J Urol. 2006;175:27–34. - PubMed
-
- Smith MR, Kabbinavar F, Saad F, Hussain A, Gittelman MC, Bilhartz DL, et al. Natural history of rising serum prostate-specific antigen in men with castrate nonmetastatic prostate cancer. J Clin Oncol. 2005;23:2918–2925. - PubMed
-
- Scher HI, Fizazi K, Saad F, Taplin ME, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–1197. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

