A phased strategy to differentiate human CD14+monocytes into classically and alternatively activated macrophages and dendritic cells
- PMID: 27401672
- PMCID: PMC5504907
- DOI: 10.2144/000114435
A phased strategy to differentiate human CD14+monocytes into classically and alternatively activated macrophages and dendritic cells
Abstract
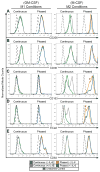
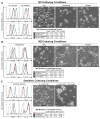
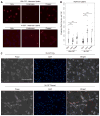
There are currently several in vitro strategies to differentiate human CD14(+) monocytes isolated from peripheral blood mononuclear cells (PBMCs) into the M1 or M2 macrophage cell types. Each cell type is then verified using flow cytometric analysis of cell-surface markers. Human CD14(+) monocytes have the potential to differentiate into M1 and M2 macrophages, both of which demonstrate varying degrees of cell-surface antigen overlap. Using multiple surface markers with current macrophage polarization protocols, our data reveal several limitations of currently used methods, such as highly ambiguous cell types that possess cell-surface marker overlap and functional similarities. Utilizing interleukin-6 (IL-6) and two phases of cytokine exposure, we have developed a protocol to differentiate human monocytes into M1, M2, or dendritic cells (DCs) with greater efficiency and fidelity relative to macrophages and DCs that are produced by commonly used methods. This is achieved via alterations in cytokine composition, dosing, and incubation times, as well as improvements in verification methodology. Our method reliably reproduces human in vitro monocyte-derived DCs and macrophage models that will aid in better defining and understanding innate and adaptive immunity, as well as pathologic states.
Keywords: M1 macrophages; M2 macrophages; cytokine; macrophage polarization; monocyte differentiation; monocyte-derived dendritic cells; multi-nucleated giant cells; peripheral blood mononuclear cells; phased strategy.
Conflict of interest statement
Figures

References
-
- Ziegler-Heitbrock L, Ancuta P, Crowe S, Dalod M, Grau V, Hart DN, Leenen PJ, Liu YJ, et al. Nomenclature of monocytes and dendritic cells in blood. Blood. 2010;116(16):e74–80. - PubMed
-
- Geissmann F, Jung S, Littman DR. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity. 2003;19(1):71–82. - PubMed
-
- Jha AK, Huang SC, Sergushichev A, Lamproupoulou V, Ivanova Y, Loginicheva E, Chmielewski K, Stewart KM, et al. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity. 2015;42(3):419–30. - PubMed
-
- Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010;11(10):889–96. - PubMed
-
- Mantovani A, Sozzani S, Locati M, Allavena P, Siva A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23(11):549–55. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
