Structure and Pharmacologic Modulation of Inhibitory Glycine Receptors
- PMID: 27401877
- PMCID: PMC4998662
- DOI: 10.1124/mol.116.105726
Structure and Pharmacologic Modulation of Inhibitory Glycine Receptors
Abstract
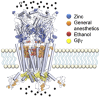
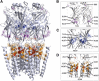
Glycine receptors (GlyR) are inhibitory Cys-loop ion channels that contribute to the control of excitability along the central nervous system (CNS). GlyR are found in the spinal cord and brain stem, and more recently they were reported in higher regions of the CNS such as the hippocampus and nucleus accumbens. GlyR are involved in motor coordination, respiratory rhythms, pain transmission, and sensory processing, and they are targets for relevant physiologic and pharmacologic modulators. Several studies with protein crystallography and cryoelectron microscopy have shed light on the residues and mechanisms associated with the activation, blockade, and regulation of pentameric Cys-loop ion channels at the atomic level. Initial studies conducted on the extracellular domain of acetylcholine receptors, ion channels from prokaryote homologs-Erwinia chrysanthemi ligand-gated ion channel (ELIC), Gloeobacter violaceus ligand-gated ion channel (GLIC)-and crystallized eukaryotic receptors made it possible to define the overall structure and topology of the Cys-loop receptors. For example, the determination of pentameric GlyR structures bound to glycine and strychnine have contributed to visualizing the structural changes implicated in the transition between the open and closed states of the Cys-loop receptors. In this review, we summarize how the new information obtained in functional, mutagenesis, and structural studies have contributed to a better understanding of the function and regulation of GlyR.
Copyright © 2016 by The American Society for Pharmacology and Experimental Therapeutics.
Figures
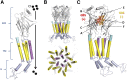
References
-
- Absalom NL, Lewis TM, Kaplan W, Pierce KD, Schofield PR. (2003) Role of charged residues in coupling ligand binding and channel activation in the extracellular domain of the glycine receptor. J Biol Chem 278:50151–50157. - PubMed
-
- Aguayo LG. (1990) Ethanol potentiates the GABAA-activated Cl− current in mouse hippocampal and cortical neurons. Eur J Pharmacol 187:127–130. - PubMed
-
- Aguayo LG, Pancetti FC. (1994) Ethanol modulation of the γ-aminobutyric acidA- and glycine-activated Cl− current in cultured mouse neurons. J Pharmacol Exp Ther 270:61–69. - PubMed
-
- Aguayo LG, Tapia JC, Pancetti FC. (1996) Potentiation of the glycine-activated Cl− current by ethanol in cultured mouse spinal neurons. J Pharmacol Exp Ther 279:1116–1122. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

