Second-Generation Non-Covalent NAAA Inhibitors are Protective in a Model of Multiple Sclerosis
- PMID: 27404798
- PMCID: PMC5009002
- DOI: 10.1002/anie.201603746
Second-Generation Non-Covalent NAAA Inhibitors are Protective in a Model of Multiple Sclerosis
Abstract
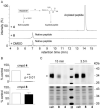
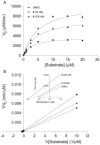
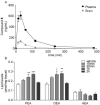
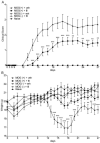
Palmitoylethanolamide (PEA) and oleoylethanolamide (OEA) are endogenous lipid mediators that suppress inflammation. Their actions are terminated by the intracellular cysteine amidase, N-acylethanolamine acid amidase (NAAA). Even though NAAA may offer a new target for anti-inflammatory therapy, the lipid-like structures and reactive warheads of current NAAA inhibitors limit the use of these agents as oral drugs. A series of novel benzothiazole-piperazine derivatives that inhibit NAAA in a potent and selective manner by a non-covalent mechanism are described. A prototype member of this class (8) displays high oral bioavailability, access to the central nervous system (CNS), and strong activity in a mouse model of multiple sclerosis (MS). This compound exemplifies a second generation of non-covalent NAAA inhibitors that may be useful in the treatment of MS and other chronic CNS disorders.
Keywords: N-acylethanolamine acid amidase; cysteine hydrolase; fatty acylethanolamides; multiple sclerosis; neuroinflammation.
© 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures
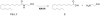
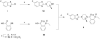
References
-
- Pontis S, Ribeiro A, Sasso O, Piomelli D. Crit Rev Biochem Mol Biol. 2016;51:7–14. - PubMed
-
- Armirotti A, Romeo E, Ponzano S, Mengatto L, Dionisi M, Karacsonyi C, Bertozzi F, Garau G, Tarozzo G, Reggiani A, Bandiera T, Tarzia G, Mor M, Piomelli D. ACS Med Chem Lett. 2012;3:422–426. - PMC - PubMed
- Ribeiro A, Pontis S, Mengatto L, Armirotti A, Chiurchiu V, Capurro V, Fiasella A, Nuzzi A, Romeo E, Moreno-Sanz G, Maccarrone M, Reggiani A, Tarzia G, Mor M, Bertozzi F, Bandiera T, Piomelli D. ACS Chem Biol. 2015;10:1838–1846. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

