Impact of Nasal Symptoms on the Quality of Life of Fresh Inductees in High Altitude
- PMID: 27408148
- PMCID: PMC4921601
- DOI: 10.1016/S0377-1237(08)80095-0
Impact of Nasal Symptoms on the Quality of Life of Fresh Inductees in High Altitude
Abstract
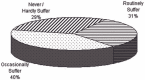
Background: Troops deployed in high altitude areas of Ladakh face nasal problems in the form of obstructive nasal symptoms and recurrent epistaxis. This study analyses the changes in the nasal mucociliary transit times (NMTT) and deterioration in quality of life of troops on first high altitude induction.
Methods: A prospective study on 100 healthy volunteers was done. Mucociliary transit time was measured and a standardized quality of life questionnaire (RQLQ) administered at onset and after a period of three months in high altitude. Thereafter the subjects were divided into two groups and the study group was administered normal saline nasal drops. The observations were taken and results were statistically analysed after a period of three months.
Result: The mucociliary transit time was prolonged after a three month stay in high altitude (p< 0.005). Normal saline nasal drops were successful in improving the nasal mucociliary transit times (p < 0.05). There was a statistically significant derangement in the quality of life which was improved by administering normal saline nasal drops (p<0.05).
Conclusion: An increased muco-cilliary transport time after exposure to high altitude denotes a deranged physiology of the nasal mucosa. This leads to a statistically significant adverse impact on the quality of life of the troops deployed. The use of normal saline nasal drops is a simple and effective method of reversing some of these changes.
Keywords: High altitude; Military medicine; Nasal mucosa.
Figures




References
-
- Barry PW, Mason NP, O'Callaghan C. Nasal mucociliary transport is impaired at altitude. Eur Respir J. 1997;10:35–37. - PubMed
-
- Salah B, Dinh Xuan AT, Fouilladieu JL, Lockhart A, Regnard J. Nasal mucociliary transport in healthy subjects is slower when breathing dry air. Eur Respir J. 1998;1:852–855. - PubMed
-
- Juniper EF, Guyatt GH. Development and testing of a new measure of health status for clinical trials in rhinoconjunctivitis. Clin Exp Allergy. 1991;21:77–83. - PubMed
-
- Juniper EF, Guyatt GH, Griffith LE, Ferrie PJ. Interpretation of rhinoconjunctivitis quality of life questionnaire data. J Allergy Clin Immunol. 1996;98:843–845. - PubMed
-
- Puchelle E, Aug F, Pham QT, Bertrand A. Comparison of three methods for measuring nasal mucociliary clearance in man. Acta Otolaryngol. 1981;91:297–303. - PubMed
LinkOut - more resources
Full Text Sources

