Physiologically Based Pharmacokinetic (PBPK) Model for Biodistribution of Radiolabeled Peptides in Patients with Neuroendocrine Tumours
- PMID: 27408897
- PMCID: PMC4938879
- DOI: 10.7508/aojnmb.2016.02.005
Physiologically Based Pharmacokinetic (PBPK) Model for Biodistribution of Radiolabeled Peptides in Patients with Neuroendocrine Tumours
Abstract
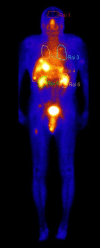
Objectives: The objectives of this work was to assess the benefits of the application of Physiologically Based Pharmacokinetic (PBPK) models in patients with different neuroendocrine tumours (NET) who were treated with Lu-177 DOTATATE. The model utilises clinical data on biodistribution of radiolabeled peptides (RLPs) obtained by whole body scintigraphy (WBS) of the patients.
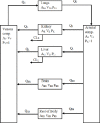
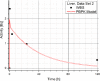
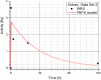
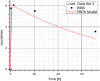
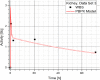
Methods: The blood flow restricted (perfusion rate limited) type of the PBPK model for biodistribution of radiolabeled peptides (RLPs) in individual human organs is based on the multi-compartment approach, which takes into account the main physiological processes in the organism: absorption, distribution, metabolism and excretion (ADME). The approach calibrates the PBPK model for each patient in order to increase the accuracy of the dose estimation. Datasets obtained using WBS in four patients have been used to obtain the unknown model parameters. The scintigraphic data were acquired using a double head gamma camera in patients with different neuroendocrine tumours who were treated with Lu-177 DOTATATE. The activity administered to each patient was 7400 MBq.
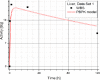
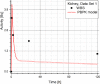
Results: Satisfactory agreement of the model predictions with the data obtained from the WBS for each patient has been achieved.
Conclusion: The study indicates that the PBPK model can be used for more accurate calculation of biodistribution and absorbed doses in patients. This approach is the first attempt of utilizing scintigraphic data in PBPK models, which was obtained during Lu-177 peptide therapy of patients with NET.
Keywords: Lu-177 DOTATATE; PBPK model; Radiolabeled peptides; Whole body scintigraphy.
Figures
Similar articles
-
Physiologically based pharmacokinetic modeling of (18)F-SiFAlin-Asp3-PEG1-TATE in AR42J tumor bearing mice.Nucl Med Biol. 2016 Apr;43(4):243-6. doi: 10.1016/j.nucmedbio.2016.01.001. Epub 2016 Jan 7. Nucl Med Biol. 2016. PMID: 27067044
-
A physiologically based pharmacokinetic (PBPK) model to describe organ distribution of 68Ga-DOTATATE in patients without neuroendocrine tumors.EJNMMI Res. 2021 Aug 16;11(1):73. doi: 10.1186/s13550-021-00821-7. EJNMMI Res. 2021. PMID: 34398356 Free PMC article.
-
Optimized Peptide Amount and Activity for ⁹⁰Y-Labeled DOTATATE Therapy.J Nucl Med. 2016 Apr;57(4):503-8. doi: 10.2967/jnumed.115.164699. Epub 2015 Dec 17. J Nucl Med. 2016. PMID: 26678617
-
An introduction to physiologically-based pharmacokinetic models.Paediatr Anaesth. 2016 Nov;26(11):1036-1046. doi: 10.1111/pan.12995. Epub 2016 Aug 23. Paediatr Anaesth. 2016. PMID: 27550716 Review.
-
Protection of Kidney Function with Human Antioxidation Protein α1-Microglobulin in a Mouse 177Lu-DOTATATE Radiation Therapy Model.Antioxid Redox Signal. 2019 May 10;30(14):1746-1759. doi: 10.1089/ars.2018.7517. Epub 2018 Aug 14. Antioxid Redox Signal. 2019. PMID: 29943622 Free PMC article. Review.
Cited by
-
Personalized metronomic radiopharmaceutical therapy through injection profile optimization via physiologically based pharmacokinetic (PBPK) modeling.Sci Rep. 2025 Feb 3;15(1):4052. doi: 10.1038/s41598-025-86159-9. Sci Rep. 2025. PMID: 39901004 Free PMC article.
-
Physiologically based radiopharmacokinetic (PBRPK) modeling to simulate and analyze radiopharmaceutical therapies: studies of non-linearities, multi-bolus injections, and albumin binding.EJNMMI Radiopharm Chem. 2024 Jan 22;9(1):6. doi: 10.1186/s41181-023-00236-w. EJNMMI Radiopharm Chem. 2024. PMID: 38252191 Free PMC article.
-
Current use and future potential of (physiologically based) pharmacokinetic modelling of radiopharmaceuticals: a review.Theranostics. 2022 Nov 14;12(18):7804-7820. doi: 10.7150/thno.77279. eCollection 2022. Theranostics. 2022. PMID: 36451855 Free PMC article. Review.
-
Mathematical Modeling of the Gastrointestinal System for Preliminary Drug Absorption Assessment.Bioengineering (Basel). 2024 Aug 9;11(8):813. doi: 10.3390/bioengineering11080813. Bioengineering (Basel). 2024. PMID: 39199771 Free PMC article.
-
A Pharmacokinetic Model Determination of Time Activity Curves in Radiopharmaceutical Therapy.Mol Imaging. 2024 Nov 3;23:15353508241280015. doi: 10.1177/15353508241280015. eCollection 2024 Jan-Dec. Mol Imaging. 2024. PMID: 40098749 Free PMC article.
References
-
- Theil FP, Guentert TW, Haddad S, Poulin P. Utility of physiologically based pharmacokinetic models to drug development and rational drug discovery candidate selection. Toxicol Lett. 2003;138(1-2):29–49. - PubMed
-
- Lee HA, Leavens TL, Mason SE, Monteiro-Riviere NA, Riviere JE. Comparison of quantum dot biodistribution with a blood-flow-limited physiologically based pharmacokinetic model. Nano Lett. 2009;9(2):794–9. - PubMed
-
- Baxter LT, Zhu H, Mackensen DG, Butler WF, Jain RK. Biodistribution of monoclonal antibodies:scale-up from mouse to human using a physiologically based pharmacokinetic model. Cancer Res. 1995;55(20):4611–22. - PubMed
-
- Baxter LT, Zhu H, Mackensen DG, Jain RK. physiologically based pharmacokinetic model for specific and nonspecific monoclonal antibodies and fragments in normal tissues and human tumor xenografts in nude mice. Cancer Res. 1994;54(6):1517–28. - PubMed
-
- Adams JC, Dills RL, Morgan MS, Kalman DA, Pierce CH. A physiologically based toxicokinetic model of inhalation exposure to xylenes in Caucasian men. Regul Toxicol Pharmacol. 2005;43(2):203–14. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
