Continuous On-Chip Cell Separation Based on Conductivity-Induced Dielectrophoresis with 3D Self-Assembled Ionic Liquid Electrodes
- PMID: 27409352
- PMCID: PMC5497574
- DOI: 10.1021/acs.analchem.6b02104
Continuous On-Chip Cell Separation Based on Conductivity-Induced Dielectrophoresis with 3D Self-Assembled Ionic Liquid Electrodes
Abstract
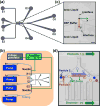
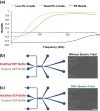
Dielectrophoresis (DEP) has been widely explored to separate cells for various applications. However, existing DEP devices are limited by the high cost associated with the use of noble metal electrodes, the need of high-voltage electric field, and/or discontinuous separation (particularly for devices without metal electrodes). We developed a DEP device with liquid electrodes, which can be used to continuously separate different types of cells or particles based on positive DEP. The device is made of polydimethylsiloxane (PDMS), and ionic liquid is used to form the liquid electrodes, which has the advantages of low cost and easy fabrication. Moreover, the conductivity gradient is utilized to achieve the DEP-based on-chip cell separation. The device was used to separate polystyrene microbeads and PC-3 human prostate cancer cells with 94.7 and 1.2% of the cells and microbeads being deflected, respectively. This device is also capable of separating live and dead PC-3 cancer cells with 89.8 and 13.2% of the live and dead cells being deflected, respectively. Moreover, MDA-MB-231 human breast cancer cells could be separated from human adipose-derived stem cells (ADSCs) using this device with high purity (81.8 and 82.5% for the ADSCs and MDA-MB-231 cells, respectively). Our data suggest the great potential of cell separation based on conductivity-induced DEP using affordable microfluidic devices with easy operation.
Figures


References
-
- Armstrong DW, Schneiderheinze JM, Kullman JP, He LF. Fems Microbiol Lett. 2001;194:33–37. - PubMed
-
- Cabrera CR, Yager P. Electrophoresis. 2001;22:355–362. - PubMed
-
- Fizazi K, Morat L, Chauveinc L, Prapotnich D, De Crevoisier R, Escudier B, Cathelineau X, Rozet F, Vallancien G, Sabatier L, Soria JC. Ann Oncol. 2007;18:518–521. - PubMed
-
- Girod M, Armstrong DW. Electrophoresis. 2002;23:2048–2056. - PubMed
-
- Hayes DF, Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Miller MC, Matera J, Allard WJ, Doyle GV, Terstappen LWWM. Clin Cancer Res. 2006;12:4218–4224. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

