Role of α7-nicotinic acetylcholine receptor in nicotine-induced invasion and epithelial-to-mesenchymal transition in human non-small cell lung cancer cells
- PMID: 27409670
- PMCID: PMC5312305
- DOI: 10.18632/oncotarget.10498
Role of α7-nicotinic acetylcholine receptor in nicotine-induced invasion and epithelial-to-mesenchymal transition in human non-small cell lung cancer cells
Abstract
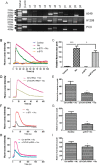
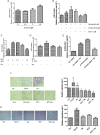
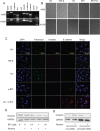
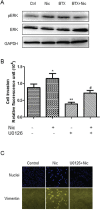
Nicotine via nicotinic acetylcholine receptors (nAChRs) stimulates non-small cell lung cancer (NSCLC) cell invasion and epithelial to mesenchymal transition (EMT) which underpin the cancer metastasis. However, the receptor subtype-dependent effects of nAChRs on NSCLC cell invasion and EMT, and the signaling pathway underlying the effects remain not fully defined. We identified that nicotine induced NSCLC cell invasion, migration, and EMT; the effects were suppressed by pharmacological intervention using α7-nAChR selective antagonists or by genetic intervention using α7-nAChR knockdown via RNA inference. Meanwhile, nicotine induced activation of MEK/ERK signaling in NSCLC cells; α7-nAChR antagonism or MEK/ERK signaling pathway inhibition suppressed NSCLC cell invasion and EMT marker expression. These results indicate that nicotine induces NSCLC cell invasion, migration, and EMT; the effects are mediated by α7-nAChRs and involve MEK/ERK signaling pathway. Delineating the effect of nicotine on the NSCLC cell invasion and EMT at receptor subtype level would improve the understanding of cancer biology and offer potentials for the exploitation of selective ligands for the control of the cancer metastasis.
Keywords: EMT; NSCLC; invasion; nicotine; α7-nAChR.
Conflict of interest statement
We declare no conflict of interest or financial disclosure.
Figures
Similar articles
-
Nicotine-Mediated Regulation of Nicotinic Acetylcholine Receptors in Non-Small Cell Lung Adenocarcinoma by E2F1 and STAT1 Transcription Factors.PLoS One. 2016 May 26;11(5):e0156451. doi: 10.1371/journal.pone.0156451. eCollection 2016. PLoS One. 2016. PMID: 27228072 Free PMC article.
-
Migration of gastric cancer is suppressed by recombinant Newcastle disease virus (rL-RVG) via regulating α7-nicotinic acetylcholine receptors/ERK- EMT.BMC Cancer. 2019 Oct 22;19(1):976. doi: 10.1186/s12885-019-6225-9. BMC Cancer. 2019. PMID: 31640627 Free PMC article.
-
Anti-tumoral activity of the human-specific duplicated form of α7-nicotinic receptor subunit in tobacco-induced lung cancer progression.Lung Cancer. 2019 Feb;128:134-144. doi: 10.1016/j.lungcan.2018.12.029. Epub 2018 Dec 28. Lung Cancer. 2019. PMID: 30642446
-
The role of α7-nAChR-mediated PI3K/AKT pathway in lung cancer induced by nicotine.Sci Total Environ. 2024 Feb 20;912:169604. doi: 10.1016/j.scitotenv.2023.169604. Epub 2023 Dec 27. Sci Total Environ. 2024. PMID: 38157907 Review.
-
Non-coding RNAs as emerging regulators of epithelial to mesenchymal transition in non-small cell lung cancer.Oncotarget. 2017 May 30;8(22):36787-36799. doi: 10.18632/oncotarget.16375. Oncotarget. 2017. PMID: 28415568 Free PMC article. Review.
Cited by
-
Contribution of the α5 nAChR Subunit and α5SNP to Nicotine-Induced Proliferation and Migration of Human Cancer Cells.Cells. 2023 Aug 4;12(15):2000. doi: 10.3390/cells12152000. Cells. 2023. PMID: 37566079 Free PMC article.
-
Treatment of Human Placental Choriocarcinoma Cells with Formaldehyde and Benzene Induced Growth and Epithelial Mesenchymal Transition via Induction of an Antioxidant Effect.Int J Environ Res Public Health. 2017 Jul 29;14(8):854. doi: 10.3390/ijerph14080854. Int J Environ Res Public Health. 2017. PMID: 28758930 Free PMC article.
-
Cell signaling and epigenetic regulation of nicotine-induced carcinogenesis.Environ Pollut. 2024 Mar 15;345:123426. doi: 10.1016/j.envpol.2024.123426. Epub 2024 Jan 29. Environ Pollut. 2024. PMID: 38295934 Free PMC article. Review.
-
α7 nicotinic acetylcholine receptors in lung cancer.Oncol Lett. 2018 Aug;16(2):1375-1382. doi: 10.3892/ol.2018.8841. Epub 2018 May 30. Oncol Lett. 2018. PMID: 30008813 Free PMC article. Review.
-
Alpha7 nicotinic acetylcholine receptor expression in Sorafenib-resistant Hepatocellular carcinoma cells.Med Oncol. 2022 Aug 16;39(11):165. doi: 10.1007/s12032-022-01745-5. Med Oncol. 2022. PMID: 35972579
References
-
- DeSantis CE, Lin CC, Mariotto AB, Siegel RL, Stein KD, Kramer JL, Alteri R, Robbins AS, Jemal A. Cancer treatment and survivorship statistics 2014. CA Cancer J Clin. 2014;64:252–271. - PubMed
-
- Edwards BK, Noone AM, Mariotto AB, Simard EP, Boscoe FP, Henley SJ, Jemal A, Cho H, Anderson RN, Kohler BA, Eheman CR, Ward EM. Annual Report to the Nation on the status of cancer 1975-2010 featuring prevalence of comorbidity and impact on survival among persons with lung colorectal breast or prostate cancer. Cancer. 2014;120:1290–1314. - PMC - PubMed
-
- Inamura K, Ishikawa Y. Lung cancer progression and metastasis from the prognostic point of view. Clin Exp Metastasis. 2010;27:389–397. - PubMed
-
- Dresler C. The changing epidemic of lung cancer and occupational and environmental risk factors. Thorac Surg Clin. 2013;23:113–122. - PubMed
-
- Cardinale A, Nastrucci C, Cesario A, Russo P. Nicotine: specific role in angiogenesis proliferation and apoptosis. Crit Rev Toxicol. 2012;42:68–89. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

