Olanzapine for the Prevention of Chemotherapy-Induced Nausea and Vomiting
- PMID: 27410922
- PMCID: PMC5344450
- DOI: 10.1056/NEJMoa1515725
Olanzapine for the Prevention of Chemotherapy-Induced Nausea and Vomiting
Abstract
Background: We examined the efficacy of olanzapine for the prevention of nausea and vomiting in patients receiving highly emetogenic chemotherapy.
Methods: In a randomized, double-blind, phase 3 trial, we compared olanzapine with placebo, in combination with dexamethasone, aprepitant or fosaprepitant, and a 5-hydroxytryptamine type 3-receptor antagonist, in patients with no previous chemotherapy who were receiving cisplatin (≥70 mg per square meter of body-surface area) or cyclophosphamide-doxorubicin. The doses of the three concomitant drugs administered before and after chemotherapy were similar in the two groups. The two groups received either 10 mg of olanzapine orally or matching placebo daily on days 1 through 4. Nausea prevention was the primary end point; a complete response (no emesis and no use of rescue medication) was a secondary end point.
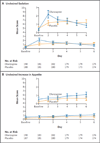
Results: In the analysis, we included 380 patients who could be evaluated (192 assigned to olanzapine, and 188 to placebo). The proportion of patients with no chemotherapy-induced nausea was significantly greater with olanzapine than with placebo in the first 24 hours after chemotherapy (74% vs. 45%, P=0.002), the period from 25 to 120 hours after chemotherapy (42% vs. 25%, P=0.002), and the overall 120-hour period (37% vs. 22%, P=0.002). The complete-response rate was also significantly increased with olanzapine during the three periods: 86% versus 65% (P<0.001), 67% versus 52% (P=0.007), and 64% versus 41% (P<0.001), respectively. Although there were no grade 5 toxic effects, some patients receiving olanzapine had increased sedation (severe in 5%) on day 2.
Conclusions: Olanzapine, as compared with placebo, significantly improved nausea prevention, as well as the complete-response rate, among previously untreated patients who were receiving highly emetogenic chemotherapy. (Funded by the National Cancer Institute; ClinicalTrials.gov number, NCT02116530.).
Figures

Comment in
-
Olanzapine for Chemotherapy-Induced Nausea and Vomiting.N Engl J Med. 2016 Oct 6;375(14):1396. doi: 10.1056/NEJMc1610341. N Engl J Med. 2016. PMID: 27705262 No abstract available.
-
Olanzapine for Chemotherapy-Induced Nausea and Vomiting.N Engl J Med. 2016 Oct 6;375(14):1395-1396. doi: 10.1056/NEJMc1610341. N Engl J Med. 2016. PMID: 27705263 No abstract available.
-
Time to re-think the olanzapine dose.Lancet Oncol. 2020 Feb;21(2):189-190. doi: 10.1016/S1470-2045(19)30791-0. Epub 2019 Dec 11. Lancet Oncol. 2020. PMID: 31838012 No abstract available.
References
-
- Navari RM. Management of chemotherapy-induced nausea and vomiting: focus on newer agents and new uses for older agents. Drugs. 2013;73:249–262. - PubMed
-
- Hesketh PJ, Rossi G, Rizzi G, et al. Efficacy and safety of NEPA, an oral combination of netupitant and palonosetron, for prevention of chemotherapy-induced nausea and vomiting following highly emetogenic chemotherapy: a randomized dose-ranging pivotal study. Ann Oncol. 2014;25:1340–1346. - PMC - PubMed
-
- Aapro M, Rugo H, Rossi G, et al. A randomized phase III study evaluating the efficacy and safety of NEPA, a fixed-dose combination of netupitant and palonosetron, for prevention of chemotherapy-induced nausea and vomiting following moderately emetogenic chemotherapy. Ann Oncol. 2014;25:1328–1333. - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- M01 RR000585/RR/NCRR NIH HHS/United States
- M01 RR000034/RR/NCRR NIH HHS/United States
- U10CA180790/CA/NCI NIH HHS/United States
- UG1 CA189956/CA/NCI NIH HHS/United States
- P30 CA012197/CA/NCI NIH HHS/United States
- UG1CA189825/CA/NCI NIH HHS/United States
- UG1 CA189830/CA/NCI NIH HHS/United States
- UG1 CA189823/CA/NCI NIH HHS/United States
- M01 RR007122/RR/NCRR NIH HHS/United States
- UG1 CA189819/CA/NCI NIH HHS/United States
- CA035269/CA/NCI NIH HHS/United States
- U10 CA035269/CA/NCI NIH HHS/United States
- UG1CA189956/CA/NCI NIH HHS/United States
- P50 CA108961/CA/NCI NIH HHS/United States
- UG1CA189823/CA/NCI NIH HHS/United States
- UG1 CA189825/CA/NCI NIH HHS/United States
- U10 CA180790/CA/NCI NIH HHS/United States
- P30 CA015083/CA/NCI NIH HHS/United States
- UG1CA189830/CA/NCI NIH HHS/United States
- 5UG1CA189819/CA/NCI NIH HHS/United States
- U10 CA180867/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
